1. sketch the shape of the orbital with the quantum numbers n=3, l=0 and ml = 0 2. the sketch of th…
Published 4 weeks ago • No plays • Length 0:33
Download video MP4
Download video MP3
Similar videos
-
 0:33
0:33
which sketch represents an orbital with the following set of quantum numbers: n =3,/ = 1,and m
-
 0:33
0:33
which orbital can be described by the quantum numbers: n = 3 l = 0 ml = 0 ?
-
 47:51
47:51
quantum numbers - n, l, ml, ms & spdf orbitals
-
 12:16
12:16
quantum numbers
-
 4:25
4:25
how to determine the 4 quantum numbers from an element or a valence electron
-
 1:00:41
1:00:41
quantum numbers class 11 chemistry
-
 21:36
21:36
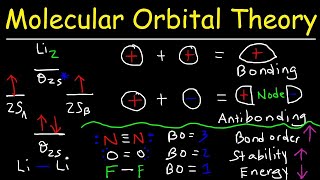
molecular orbital theory - bonding & antibonding mo - bond order
-
 11:46
11:46
how to determine the maximum number of electrons using allowed quantum numbers - 8 cases
-
 0:33
0:33
1. write the set of quantum numbers for the following a. it is an up-spin 4d electron with an orbit…
-
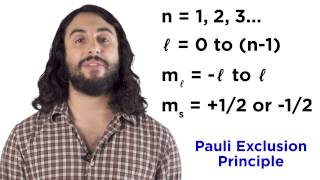 8:42
8:42
quantum numbers, atomic orbitals, and electron configurations
-
 0:33
0:33
give the name, magnetic quantum numbers, and number of orbitals for each sublevel with the followin…
-
 11:19
11:19
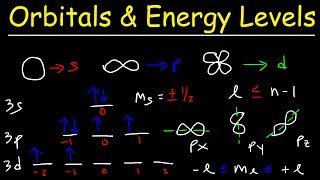
orbitals, atomic energy levels, & sublevels explained - basic introduction to quantum numbers
-
 0:33
0:33
an electron has the following set of quantum numbers: 3, 2, -1,
-
 0:33
0:33
'5 the subshell letter: a) specifies the principal shell of the orbital. b) specifies the 3-d shape…
-
 0:33
0:33
complete the diagram for the hierarchy of quantum numbers for atomic orbitals. enter all allowable …
-
 0:54
0:54
quantum numbers and atomic orbital shape
-
 22:15
22:15
2.2 part 1 - quantum numbers
-
 0:33
0:33
which of the following sets of quantum numbers (ordered n, â„“, mâ„“) are possible for an electron …
-
 0:33
0:33
which one of the following represents an acceptable set of quantum numbers for an electron in an at…
-
 1:34:06
1:34:06
quantum numbers - the easy way!
-
 0:33
0:33
questionm11 what is the maximum number of electrons in an atom that can have the following set of q…
-
 13:55
13:55
quantum numbers
Clip.africa.com - Privacy-policy
 0:33
0:33
 0:33
0:33
 47:51
47:51
 12:16
12:16
 4:25
4:25
 1:00:41
1:00:41
 21:36
21:36
 11:46
11:46
 0:33
0:33
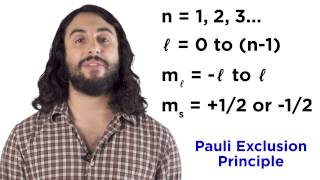 8:42
8:42
 0:33
0:33
 11:19
11:19
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:54
0:54
 22:15
22:15
 0:33
0:33
 0:33
0:33
 1:34:06
1:34:06
 0:33
0:33
 13:55
13:55