1. the zero-order reaction a → products takes 63.5 minutes for the concentration of a to decrease…
Published 10 months ago • 3 plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 48:46
48:46
integrated rate laws - zero, first, & second order reactions - chemical kinetics
-
 0:33
0:33
for a first-order reaction of the form a →n b (with n possibly fractional) and […
-
 11:34
11:34
chemical kinetics: first-order calculations. concentration and time. full video.@zulubaconsultancy
-
 11:06
11:06
concentration vs time for a zero order reaction | kinetics | chemistry | khan academy
-
 4:42
4:42
zero-order reactions | kinetics | ap chemistry | khan academy
-
 0:33
0:33
in a first-order reaction with only one reagent, the reaction was started with a concentration of r…
-
 4:06
4:06
first order kinetics calculations
-
 13:15
13:15
what is a zero order reaction ? | kinetics | chemistry | khan academy
-
 7:44
7:44
first-order reactions | kinetics | ap chemistry | khan academy
-
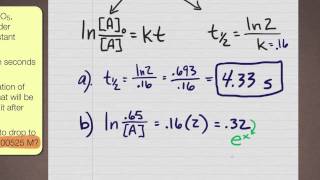 7:03
7:03
first order kinetics problem
-
 11:28
11:28
worked example - zero order reaction | kinetics | chemistry | khan academy
-
 0:33
0:33
the initial concentration of reactant in a first-order reaction is 0.89 m. the rate constant for th…
-
 0:33
0:33
for each of the following reactions, express the rate of change of concentration of the reactants a…
-
 4:38
4:38
chemistry - chemical kinetics (11 of 30) finding the order - using initial rates
-
 3:53
3:53
zero, first, and second order reactions
-
 0:33
0:33
a drug has the initial concentration of 5.0 x 10^-3 g/ml in aqueous solution. after 24 months, the …
-
 0:33
0:33
which one of the following statements does not describe the equilibrium state? a) equilibrium is dy…
-
 9:53
9:53
zero-order reaction (with calculus) | kinetics | chemistry | khan academy
-
 11:46
11:46
calculations on first order kinetics
-
 0:33
0:33
consider the reaction: 2o3 – 3o2 (a) the rate become 4 times faster if the initial o3 concentrati…
-
 5:26
5:26
zero order kinetics example problem
-
 3:30
3:30
understanding zero order and first order kinetics