14.40c | which acid is stronger? hso3− or hso4−
Published 2 years ago • 2.2K plays • Length 5:39Download video MP4
Download video MP3
Similar videos
-
 5:25
5:25
14.40d | which acid is stronger? nh3 or h2s
-
 5:12
5:12
14.40e | which acid is stronger? h2o or h2te
-
 5:39
5:39
14.38 | which is the stronger acid, nh4 or hbro?
-
 6:22
6:22
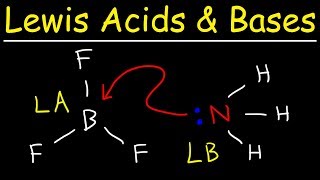
lewis acids and bases
-
 6:00
6:00
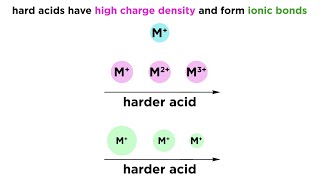
hard/soft acid-base theory
-
 11:37
11:37
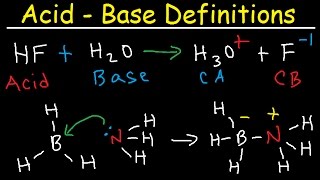
conjugate acid base pairs, arrhenius, bronsted lowry and lewis definition - chemistry
-
 6:26
6:26
14.39 | which is the stronger base, (ch3)3n or h2bo3−?
-
 10:31
10:31
15.10c | calculate the solubility product for gd2(so4)3, 3.98 g/100 ml
-
 3:45
3:45
14.3f | how does hso4− acts as a brønsted-lowry acid?
-
 4:25
4:25
14.40b | which acid is stronger? b(oh)3 or al(oh)3
-
 3:53
3:53
14.3b | how does hcl act as a brønsted-lowry acid?
-
 3:42
3:42
14.69a | is al(no3)3 acidic, basic, or neutral?
-
 4:24
4:24
14.6c | how does nh2− acts as a brønsted-lowry base?
-
 3:30
3:30
14.5c | how does nh3 acts as a brønsted-lowry base?
-
 4:15
4:15
14.6e | how does o2− acts as a brønsted-lowry base?
-
 4:29
4:29
14.9a | how to identify the conjugate acid-base pairs in hno3 h2o → h3o no3−
-
 7:25
7:25
using charge to rank acid base strength in organic chemistry