16.60a-b | the evaporation of one mole of water at 298 k has a standard free energy change of 8.58
Published 1 year ago • 1.7K plays • Length 7:29
Download video MP4
Download video MP3
Similar videos
-
 8:05
8:05
16.60c | by calculating ∆g, determine if the evaporation of water at 298 k is spontaneous when the
-
 7:45
7:45
16.60d | if the evaporation of water were always nonspontaneous at room temperature, wet laundry
-
 11:01
11:01
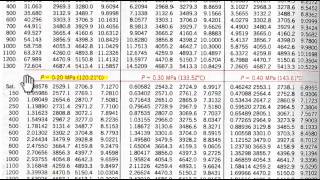
reading water tables
-
 9:13
9:13
16.45 | consider the following reaction at 298 k: n2o4(g) ⇌ 2no2(g) kp = 0.142 what is the standard
-
 5:31
5:31
16.34c | determine the free energy change for 2cu(s) s(g) → cu2s(s)
-
 5:35
5:35
16.33b | how to find the free energy change for h2(g) br2(l) → 2hbr(g)
-
 5:19
5:19
16.34a | how to find the free energy change for c(s, graphite) o2(g) → co2(g)
-
 11:03
11:03
16.58 | determine the standard free energy change, δg°f , for the formation of s2−(aq) given that
-
 4:15
4:15
digital water flow rate balancing meter | eco climate solutions
-
 9:24
9:24
14.86 | what will be the ph of a buffer solution prepared from 0.20 mol nh3, 0.40 mol nh4no3, and
-
 4:59
4:59
actiflo® carb, optimal treatment of natural organic matter
-
 5:22
5:22
16.34b | how to determine the free energy change for o2(g) n2(g) → 2no(g)
-
 7:22
7:22
16.34e | determine the free energy change for fe2o3(s) 3co(g) → 2fe(s) 3co2(g)
-
 9:23
9:23
what is a mole? it's chemistry's best friend!
-
 11:39
11:39
16.59 | determine the standard enthalpy change, entropy change, and free energy change for the
-
 11:25
11:25
steam reformer material and energy balance
-
 6:41
6:41
16.34f | find the free energy change for caso4·2h2o(s) → caso4(s) 2h2o(g)
-
 42:04
42:04
everything you need to know about the ideal gas law
-
 3:36
3:36
16.30 | what is the difference between δg and δg° for a chemical change?
-
 8:02
8:02
16.64 | when ammonium chloride is added to water and stirred, it dissolves spontaneously and the
-
 6:12
6:12
crystallization calculations - solve crystallization problems - crystallization by cooling
-
 6:12
6:12
evaporative cooling energy balance
Clip.africa.com - Privacy-policy
 8:05
8:05
 7:45
7:45
 11:01
11:01
 9:13
9:13
 5:31
5:31
 5:35
5:35
 5:19
5:19
 11:03
11:03
 4:15
4:15
 9:24
9:24
 4:59
4:59
 5:22
5:22
 7:22
7:22
 9:23
9:23
 11:39
11:39
 11:25
11:25
 6:41
6:41
 42:04
42:04
 3:36
3:36
 8:02
8:02
 6:12
6:12
 6:12
6:12