17.31a | calculate the equilibrium constant for agcl(s) ⇌ ag (aq) cl−(aq) using cell potentials
Published 1 year ago • 5.6K plays • Length 11:49Download video MP4
Download video MP3
Similar videos
-
 10:48
10:48
equilibrium constant k & cell potential problems with ksp - electrochemistry
-
 5:35
5:35
16.39f | calculate δg° from the equilibrium constant: agbr(s) → ag (aq) br−(aq) kp = 2.2×10^−13
-
 15:43
15:43
16.43c | calculate the equilibrium constant for 2lioh(s) co2(g) → li2co3(s) h2o(g) (t = 575 °c)
-
 5:11
5:11
16.40c | how to calculate δg° from the equilibrium constant: h2o(l) ⇌ h2o(g) t = 60°c kp = 0.196
-
 9:43
9:43
calculating the equilibrium constant from the standard cell potential | khan academy
-
 7:55
7:55
13.10b | write the expression for the equilibrium constant for the reaction represented by the
-
 9:27
9:27
16.57 | given that the δg°f for pb2 (aq) and cl−(aq) is −24.3 kj/mole and −131.2 kj/mole
-
 1:27:17
1:27:17
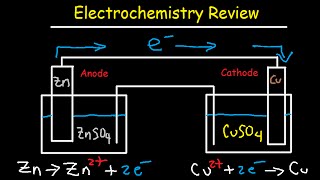
electrochemistry review - cell potential & notation, redox half reactions, nernst equation
-
 30:53
30:53
nernst equation explained, electrochemistry, example problems, ph, chemistry, galvanic cell
-
 34:06
34:06
how to balance redox reactions - general chemistry practice test / exam review
-
 5:22
5:22
standard cell potential and the equilibrium constant | chemistry | khan academy
-
 10:56
10:56
cell potential problems - electrochemistry
-
 6:29
6:29
17.21a | calculate the standard cell potential for mg(s) ni2 (aq) → mg2 (aq) ni(s)
-
 2:01
2:01
13.21d | is baso3(s) ⇌ bao(s) so2(g) at a homogeneous or a heterogeneous equilibrium?
-
 30:20
30:20
13.82 | the equilibrium constant (kc) for this reaction is 1.60 at 990 °c: h2(g) co2(g) ⇌ h2o(g)
-
 4:59
4:59
13.30 | what is the approximate value of the equilibrium constant kp for the changec2h5oc2h5(l) ⇌
-
 6:23
6:23
15.13a | calculate the molar solubility of khc4h4o6 from its solubility product