2.24 | variations in average atomic mass may be observed for elements obtained from different
Published 4 years ago • 2.5K plays • Length 9:52Download video MP4
Download video MP3
Similar videos
-
 5:27
5:27
2.23 | average atomic masses listed by iupac are based on a study of experimental results. bromine
-
 5:07
5:07
6.71 | rank the following atoms in order of increasing first ionization energy: f, li, n, rb
-
 14:10
14:10
6.47 | astronomical observations of our milky way galaxy indicate that it has a mass of about
-
 18:03
18:03
chemistry practice problems: atomic mass calculations ii
-
 15:44
15:44
4.20e | identify the atoms that are oxidized and reduced: 2k2s2o3(s) i2(s) → k2s4o6(s) 2ki(s)
-
 1:15:40
1:15:40
22. quantum mechanics iv: measurement theory, states of definite energy
-
 10:20
10:20
2.12 | write the symbol for each of the following ions: (a) the ion with a 3 charge, 28 electrons
-
 3:14
3:14
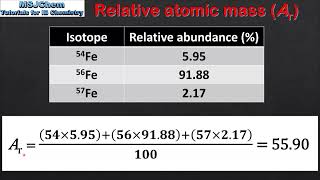
s1.2.2 calculating relative atomic mass
-
 8:41
8:41
11.27 | what is the microscopic explanation for the macroscopic behavior illustrated in figure
-
 6:03
6:03
13.40 | average atomic and molecular speeds (vrms) are large, even at low temperatures. what is vrms
-
 7:20
7:20
closer look: calculating average atomic mass | chemistry matters
-
 10:19
10:19
average atomic mass practice problems
-
 6:32
6:32
2.17 | the following are properties of isotopes of two elements that are essential in our diet.
-
 7:27
7:27
13.61d | complete the changes in pressure for 2nh3(g) 2o2(g) ⇌ n2o(g) 3h2o(g)
-
 7:46
7:46
9.64 | in an experiment in a general chemistry laboratory, a student collected a sample of a gas
-
 8:52
8:52
2.6 | how are electrons and protons similar? how are they different?
-
 9:21
9:21
15.10a | calculate the solubility product for basif6, 0.026 g/100 ml (contains sif6 2− ions)
-
 15:27
15:27
2.11 | write the symbol for each of the following ions: (a) the ion with a 1 charge, atomic number
-
 8:00
8:00
2.7 | how are protons and neutrons similar? how are they different?
-
 9:26
9:26
2.35 | use the build a molecule simulation (http://openstax.org/l/16molbuilding) to repeat exercise
-
 5:23
5:23
14.53a | how to calculate the ka for ch3co2h from equilibrium concentrations