250 ml of a solution contains 6.3 grams of oxalic acid (mol. wt. =126 g / mol ). what is the vol...
Published 1 year ago • 1.3K plays • Length 2:27Download video MP4
Download video MP3
Similar videos
-
 7:18
7:18
if \(250 \mathrm{~cm}^3\) of an aqueous solution containing \(0.73 \mathrm{~g}\) of a protein \(....
-
 9:52
9:52
15.16a | calculate the concentration of all solute species: tlcl(s) in 1.250 m hcl
-
 3:23
3:23
how to determine the amount of solution that contains a given amount of solute
-
 4:48
4:48
an aqueous solution of \( 6.3 \mathrm{~g} \) oxalic acid dihydrate ...
-
 4:41
4:41
calculating grams when making a solution from a solid
-
 3:42
3:42
solute, solvent and solution | what is a solution? | science video for kids
-
 12:06
12:06
dosage calculations made easy | reconstitution calculation medication problems nursing students (10)
-
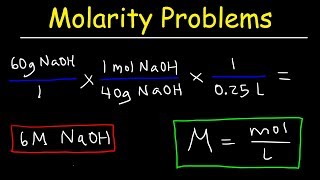 21:27
21:27
molarity practice problems
-
 6:01
6:01
250 ml of a calcium carbonate solution contains 2.5 grams of caco_(3). if 10 ml of this solution...
-
 3:29
3:29
the concentration of a \( 100 \mathrm{ml} \) solution containing \(...
-
 0:28
0:28
how to calculate % solution - biology and chemistry 🧪 #solutions #chemistry #college
-
 31:25
31:25
molarity, molality, volume & mass percent, mole fraction & density - solution concentration problems
-
 3:25
3:25
the molarity of a 250 ml solution is 0.5 m. the amount of solute present in it is g. (molecular ...
-
 2:44
2:44
the volume of water that must be added to a mixture of \( 250 \math...
-
 3:23
3:23
calculate %v/v (percent by volume of a solution)
-
 5:05
5:05
, an aqueous solution of 6.3 gm of oxalic acid dihydrate is made upto 250 ml. the volume of 0.1 ...
-
 0:06
0:06
q61,the molarity of a sol obtained by mixing 750 ml of 0.5 m hcl with 250 ml of 2 m hcl will be
-
 0:59
0:59
the osmotic pressure of solution containing 4.0g of solute (molar mass 246) per litre at 27
-
 0:54
0:54
250 ml of a sodium carbonate solution contains 2.65 grams of na2co3. #gauravsir #study #india
-
 3:25
3:25
the molarity of a solution obtained by mixing 750 ml of 0.5(m) hcl with 250 ml of 2(m) hcl will ...
-
 3:46
3:46
an aqueous solution of 6.3 g oxalic acid dihydrate is made up to 250 ml. the volume of 0.1 nna...
-
![the freezing point (in `.^(@)c)` of a solution containing `0.1g` of `k_(3)[fe(cn)_(6)]` (mol. wt.](https://i.ytimg.com/vi/DKjm_tFSZ9U/mqdefault.jpg) 2:53
2:53
the freezing point (in `.^(@)c)` of a solution containing `0.1g` of `k_(3)[fe(cn)_(6)]` (mol. wt.