4.37d | balance the half-reaction: h2o(l) → o2(g) (in acidic solution)
Published 2 years ago • 4K plays • Length 8:43Download video MP4
Download video MP3
Similar videos
-
 8:32
8:32
4.37g | balance the half-reaction: mno4−(aq) → mn2 (aq) (in acidic solution)
-
 7:31
7:31
balancing redox reactions in acidic and basic conditions
-
 16:00
16:00
half reaction method, balancing redox reactions in basic & acidic solution, chemistry
-
 15:00
15:00
how to balance redox equations in acidic solution
-
 12:30
12:30
4.37h | balance the half-reaction: cl−(aq) → clo3−(aq) (in basic solution)
-
 7:52
7:52
4.37f | balance the half-reaction: so32−(aq) → so42−(aq) (in acidic solution)
-
 9:01
9:01
4.38f | balance: no3−(aq) → hno2(aq) (in acidic solution)
-
 6:09
6:09
4.37a | balance the half-reaction: sn4 (aq) → sn2 (aq)
-
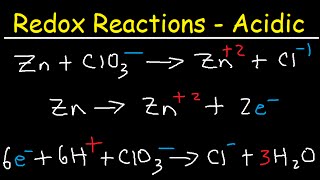 20:13
20:13
how to balance redox equations in acidic solution
-
 15:45
15:45
how to balance redox equations in basic solution
-
 11:41
11:41
redox reactions
-
 8:57
8:57
4.38h | balance: cl−(aq) → clo3−(aq) (in acidic solution)
-
 15:36
15:36
4.40e | balance: h2o2(aq) mno4−(aq) → mn2 (aq) o2(g) (in acid)
-
 25:15
25:15
balancing a redox equation in acidic solution
-
 4:29
4:29
balance redox reaction (ionic half equation method)
-
 14:49
14:49
17.4b | balance the equation in an acidic solution: pbo2 hg → hg22 pb2
-
 3:11
3:11
how to balance co(g) o2(g) → co2(g)
-
 3:42
3:42
r3.2.2 balancing redox equations in acidic solutions
-
 13:37
13:37
4.39e | balance: hbro(aq) → br−(aq) o2(g) (in acid)
-
 5:13
5:13
balance redox reaction: acidic solution 2 (example)
-
 8:55
8:55
balancing redox reaction example
-
 14:46
14:46
how to balance redox equations in acidic solution example 1