5 section 7.3 iso 13485 design & development - 7.3
Published 3 years ago • 661 plays • Length 11:26Download video MP4
Download video MP3
Similar videos
-
 10:17
10:17
7 section 7.5 iso 13485 product and service provision - 7.5
-
 4:31
4:31
risk management 820.30g & iso 13485 § 7.1, 7.3.3, & 7.3.9 (executive series #21)
-
 5:23
5:23
understanding quality management systems - iso 13485 - clause 7.3 - design & development
-
 5:08
5:08
why you need iso 13485 for your medical device manufacturing project
-
 13:11
13:11
iso 13485: what you need to know to build a quality management systems for medical devices
-
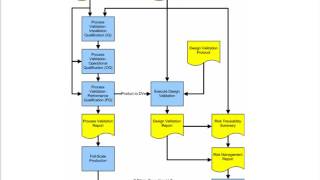 1:28:00
1:28:00
process validation for medical device manufacturers
-
 3:52
3:52
steam sterilization iso 13485 § 7.5.7 (executive series #85)
-
 5:18
5:18
sterilization overkill methods iso 13485 § 7.5.6 & 7.5.7 (executive series #93)
-
 3:59
3:59
sterilization families iso 13485 § 7.5.6 & 7.5.7 (executive series 96)
-
 0:32
0:32
why iso 13485?
-
 3:46
3:46
design planning 820.30b & iso 13485 § 7.3.2 (executive series #11)
-
 2:55
2:55
understanding quality management systems - iso 13485 - clause 7.1 - planning of product realization
-
 3:22
3:22
design inputs 820.30c & iso 13485 § 7.3.3 (executive series #12)
-
 3:51
3:51
design review 820.30e & iso 13485 § 7.3.5 (executive series #14)
-
 11:26
11:26
iso 13485 design
-
 4:02
4:02
sterilization revalidation – iso § 7.5.6 and 7.5.7 (executive series #95)
-
 3:47
3:47
design validation 820.30g & iso 13485 § 7.3.7 (executive series #16)
-
 4:45
4:45
understanding quality management systems - iso 13485 - clause 5.3 - quality policy
-
 3:29
3:29
design verification 820.30f & iso 13485 § 7.3.6 (executive series #15)
-
 11:12
11:12
what is iso 13485?