6.66 | write a set of quantum numbers for each of the electrons with an n of 3 in a sc atom.
Published 4 years ago • 2.8K plays • Length 8:43
Download video MP4
Download video MP3
Similar videos
-
 4:25
4:25
how to determine the 4 quantum numbers from an element or a valence electron
-
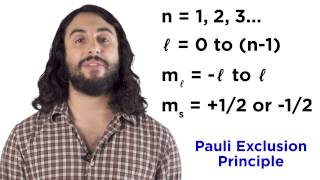 8:42
8:42
quantum numbers, atomic orbitals, and electron configurations
-
 4:03
4:03
quantum numbers - assigning the electrons in a nitrogen atom
-
 12:16
12:16
quantum numbers
-
 11:46
11:46
how to determine the maximum number of electrons using allowed quantum numbers - 8 cases
-
 4:38
4:38
6.73 | atoms of which group in the periodic table have a valence shell electron configuration of ns2
-
 14:17
14:17
how to determine the maximum number of electrons given a set of quantum numbers
-
 12:01
12:01
spdf orbitals explained - 4 quantum numbers, electron configuration, & orbital diagrams
-
 5:50
5:50
atomic orbitals 3d
-
 10:52
10:52
orbitals: crash course chemistry #25
-
 14:28
14:28
orbitals, the basics: atomic orbital tutorial — probability, shapes, energy |crash chemistry academy
-
 4:11
4:11
6.35 | identify the subshell in which electrons with the following quantum numbers are found
-
 6:26
6:26
6.34 | answer the following questions: (a) without using quantum numbers, describe the differences
-
 7:20
7:20
quantum numbers - how many electrons and orbitals have the following set of quantum numbers?
-
 1:34:06
1:34:06
quantum numbers - the easy way!
-
 38:44
38:44
orbitals, quantum numbers & electron configuration - multiple choice practice problems
-
 14:08
14:08
aleks - deducing the allowed quantum numbers of an atomic electron
-
 7:26
7:26
6.43 | how many electrons could be held in the second shell of an atom if the spin quantum number ms
-
 12:00
12:00
quantum numbers | electronic structure of atoms | chemistry | khan academy
-
 47:51
47:51
quantum numbers - n, l, ml, ms & spdf orbitals
-
 5:43
5:43
6.32 | what are the allowed values for each of the four quantum numbers: n, l, ml, and ms?
-
 21:15
21:15
quantum numbers for electrons
Clip.africa.com - Privacy-policy
 4:25
4:25
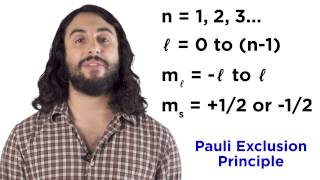 8:42
8:42
 4:03
4:03
 12:16
12:16
 11:46
11:46
 4:38
4:38
 14:17
14:17
 12:01
12:01
 5:50
5:50
 10:52
10:52
 14:28
14:28
 4:11
4:11
 6:26
6:26
 7:20
7:20
 1:34:06
1:34:06
 38:44
38:44
 14:08
14:08
 7:26
7:26
 12:00
12:00
 47:51
47:51
 5:43
5:43
 21:15
21:15