a 2.5 l flask at 15â°c contains a mixture of n2, he, and ne gases with the partial pressure of 0.32…
Published 2 months ago • 4 plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 0:33
0:33
two flasks, a 1.0 l flask containing no gas and a 1.5 l flask containing n2o4 gas, are maintained a…
-
 0:33
0:33
when 2.00 mol of so2cl2 is placed in a 2.00 l flask at 303 k, 56
-
 0:33
0:33
be sure to answer all parts. three 5-l flasks, fixed with pressure gauges and small valves, each co…
-
 0:33
0:33
. find the pressure in mm hg produced by 2.35 g of carbon dioxide in a 5.00 l flask at 18oc.
-
 0:33
0:33
a 1.5 l flask is filled with nitrogen at a pressure of 12 atmospheres. what size flask would be req…
-
 0:33
0:33
a 2.00- l flask is filled with ethane gas (c_2 h_6) from a small cylinder, as shown …
-
 0:33
0:33
how many molecules of n2 gas are present in a 2.5-l flask at 50*c and 650 torr? 4.9 * 1022 molecule…
-
 0:33
0:33
a 1-l flask is filled with 1.45 g of argon at 25 â°c. a sample of ethane vapor is added to the same…
-
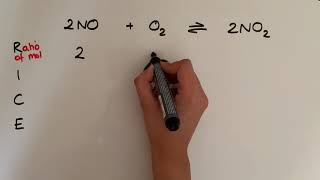 10:13
10:13
using rice to calculate equilibrium concentrations
-
 53:22
53:22
chemical equilibrium constant k - ice tables - kp and kc
-
 17:34
17:34
a levels chemistry.ll feb/mar 19 p42 ll question no 6 (mass spectroscopy)
-
 0:33
0:33
0.257 mol/l of a2(g) and 0.257 mol/l b2(g) are placed in a flask. a2 and b2 react according to the …
-
 0:33
0:33
assume that the initial purple solution is 16.05 mg/l in sodium salicylate, 0.15 m in hcl, and 0.06…
-
 0:33
0:33
what is the temperature of a 1.59 l flask containing 0.175 mol of ne at a pressure of 1.09 atm?
-
 0:33
0:33
question 1 (1 point) saved when sealed 1l flask of gas is cooled down; what happens to the gas mole…
-
 0:33
0:33
equilibrium pressure at a particular temperature, kp = 0.230 for the reaction: n2o4(g) ⇜ 2no2(g) …
-
 0:33
0:33
the pressure in a 30.0 l flask is 0.422 atm at 82 â°c. how many moles of gas are in the flask? nat…
-
 0:33
0:33
use the ideal gas law to determine the molar mass (g/mol) of an unknown gas in a 152 ml flask at 65…
-
 0:33
0:33
a flask of unknown volume containing a gas at 4.0 atm is attached to a 5.0 l evacuated flask. the a…
-
 0:33
0:33
an empty flask has a mass of 47.392 g and 47.816 g when filled with acetone vapor at 100.0â°c and 7…
-
 0:33
0:33
hydrogen gas can be generated from the rcaction between aluminum metal and hydrochloric acid: 2 al …
-
 0:33
0:33
calculate the total pressure (in atm) of the mixture when 1.00 atm of dinitrogen reacts with 5.00 a…