a solution of 49 0% h2so4 by mass has a density of 1 39 gcm3 at 293 k a 25 0 cm3 sample of this sol
Published 1 month ago • 10 plays • Length 3:11Download video MP4
Download video MP3
Similar videos
-
 3:48
3:48
2.00 m h2so4 has a density of 1.15 g/ml. what is the % by mass of solute in this solution?
-
 0:33
0:33
a sulfuric acid solution containing 571.4 g of h2so4 per liter of solution has a density of 1.329 g…
-
 2:54
2:54
`49 g` of `h_(2) so_(4)` is disslved in enough water to make one litre of a soltuion of density `
-
 0:33
0:33
a 9.386 m aqueous solution of sulfuric acid has a density of 1.5091 g cm-3. calculate the molality,…
-
 31:25
31:25
molarity, molality, volume & mass percent, mole fraction & density - solution concentration problems
-
 4:11
4:11
how to prepare 0.1 m naoh solution?|| calculations and experiment
-
 3:36
3:36
how to calculate density - with examples
-
 21:27
21:27
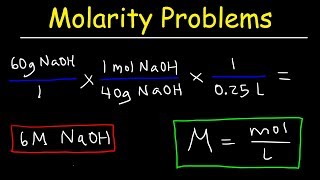
molarity practice problems
-
 3:04
3:04
a solution of sulphuric acid in water that is `25% h_(2)so_(4)` by mass has a density of `1.178gml
-
 3:45
3:45
a 50.0 % (w/w) solution of sulfuric acid in water has a density of 1.395 g/ml. what is its molarity?
-
 4:29
4:29
a 7.112 m solution of sulfuric acid in water has a density of 1.395 g/ml. what is its molality?
-
 1:57
1:57
a solution of h2so4 is 31.4% h2so4 by mass and has a density of 1.25g/ml. the molarity of the h2so4
-
 1:53
1:53
a solution of h2so4 is 31.4% h2so4 by mass and has a density of 1.25g/ml ii29th january ,2024 ii
-
 6:34
6:34
chemistry: calculating molarity from percent mass and density
-
 4:47
4:47
calculate morality of 10%of aqueous solution of h2so4. density of solution is 1.47 gml-¹#class12th
-
 0:21
0:21
calculate the molarity of 9.8% (w/w) solutionof h2so4--. #chemistry#class12th#pyqs#molality#molarity
-
 4:35
4:35
preparing dilute sulfuric acid from a stock solution
-
 0:33
0:33
how many moles of h2so4 are in 2.50 l of a 4.25 m aqueous solution?
-
 4:10
4:10
the density of a solution containing 13% by mass of h2so4 is 1.09 g/ml. calculate the molarity#study
-
 7:09
7:09
concentrated h2so4 has a density 1.9g/ml and is 99% h2so4 by mass. calculate the molarity.
-
 0:33
0:33
calculate the molarity of the h2so4 solution if it takes 40.0 ml of h2so4 to neutralize 0.364 g of …
-
 4:13
4:13
mass percent sulfuric acid