1.) calculate the ph of a 0.50 m solution of ammonia (nh3, kb = 1.8 x 10-5.)
Published 3 months ago • 7 plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 7:05
7:05
calculate the ph of 0.1 m acetic acid
-
 1:21
1:21
reaction of nh3 (g) hcl (g) can two gases make a solid? 🌪
-
 0:33
0:33
what is the equilibrium concentration of ammonium ion in a 0.33 m solution of ammonia (nh3, kb = 1.…
-
 1:07
1:07
gas phase reaction hcl and nh3
-
 1:07
1:07
gas phase, acid base reaction between ammonia and hydrochloric acid
-
 6:28
6:28
ph of nh3 solution (example)
-
 8:39
8:39
ph of a weak acid (0.1 m acetic acid) example
-
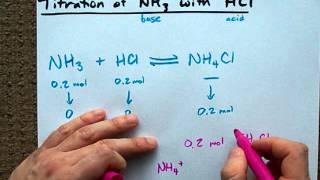 2:58
2:58
easy way to understand the nh3 with hcl titration
-
 3:05
3:05
ammonia and hydrogen chloride diffusion experiment
-
 13:51
13:51
the end of haber bosch
-
 2:14
2:14
hcl nh3=nh4cl - ammonium chloride smoke under 100,000,000x microscope
-
 3:19
3:19
equation for nh3 h2o (ammonia water)
-
 0:33
0:33
how does concentration affect ph: 1.0m nh4cl 1.0m nh4oh
-
![chemistry: what is the ph of a solution with [h ] = 1x10^-3 ?](https://i.ytimg.com/vi/rXTbF2ngoL0/mqdefault.jpg) 1:28
1:28
chemistry: what is the ph of a solution with [h ] = 1x10^-3 ?
-
 0:33
0:33
calculate the percent ionization of an acetic acid solution having the given concentration. a. 1.00…
-
 5:56
5:56
r3.1.10 calculating the ka or kb of a weak acid or base (hl)
-
 0:33
0:33
calculate the concentrations of acetic acid (p k_u=4.76) and sodium …
-
 1:26
1:26
balance nh3 hcl = nh4cl (ammonia and hydrochloric acid)
-
 4:36
4:36
18.1.4 ka and kb
-
 0:55
0:55
how does amount of strong acid/base affect change in ph: 0.1m nh4oh 0.1m nh4cl add 0.010mol hcl
-
 0:33
0:33
identify the acid, base, conjugate acid and conjugate base in the following reactions: 1. nh3(aq) …
-
 1:41
1:41
addition of hcl and naoh to a 0.10 m nh3 / nh4cl buffer - buffer activity lab part 5