biggest challenges when transitioning legacy products and how to mitigate these: medical devices
Published 3 months ago • 4 plays • Length 2:52Download video MP4
Download video MP3
Similar videos
-
 14:27
14:27
medical device design & development – challenges when transitioning legacy products
-
 1:51
1:51
medical device labelling - three biggest challenges of labelling & udi
-
 13:41
13:41
what are the biggest regulatory challenges for combination products? device perspective
-
 11:24
11:24
medical device classification overview
-
 2:53
2:53
defining critical design inputs for medical devices
-
 1:28:00
1:28:00
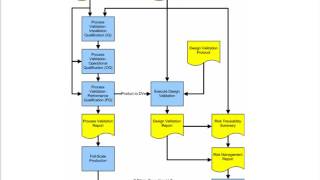
process validation for medical device manufacturers
-
 1:11:09
1:11:09
how to do software as medical device development correctly?
-
 2:24
2:24
semi automatic pvc conduit pipe bending machine.............. 91-9951742288
-
 5:32
5:32
medical device & ivd career advice - challenges of a career in medical device regulatory affairs
-
 0:41
0:41
benefits & challenges of the mdr transition to medical device manufacturers.
-
 12:03
12:03
a short guide to software as a medical device | interview with richard young
-
 9:50
9:50
a quick guide to internal audits for medical devices and ivds
-
 1:55
1:55
medical device labelling - differences between the mdr and mdd
-
 3:03
3:03
the biggest regulatory challenges when navigating the combination products landscape
-
 2:03
2:03
understanding state-of-the-art for medical device portions of combination products
-
 14:16
14:16
how to get a medical device on the market
-
 1:33
1:33
what is product and process validation and why do we have to do it?
-
 16:41
16:41
how will the ivdr impact you?
-
 3:30
3:30
5.2 research and development strategies in medical devices
-
 2:16
2:16
how test automation improves regulatory submissions in medical devices