biologics, biosimilars, and the two-fold u.s. approval framework’s possible impact on prices
Published 1 year ago • 3.6K plays • Length 32:37Download video MP4
Download video MP3
Similar videos
-
 4:12
4:12
the basics of biosimilars
-
 7:21
7:21
biologic drugs and biosimilars
-
 2:47
2:47
biosimilars: approval process
-
 4:41
4:41
biologics, unbranded biologics, and biosimilars - what is the difference?
-
 8:07
8:07
biosimilars | what is a biosimilar? | gastrointestinal society
-
 6:07
6:07
manufacturing of biologics
-
 3:00
3:00
biosimilars: complex copycat drugs
-
 3:18
3:18
what is a biosimilar, exactly?
-
 5:20
5:20
data requirements for biosimilars
-
 2:47
2:47
biologics vs. biosimilars: what’s the difference?
-
 14:19
14:19
regulatory pathway for biosimilars
-
 38:15
38:15
biosimilars and biologics
-
 0:25
0:25
the power of biosimilars: the benefits of fda-approved biologics #shorts
-
 1:36
1:36
interchangeable biosimilars
-
 2:04
2:04
the impact of the fda's approval of the first 'biosimilar' drug
-
 6:54
6:54
the biosimilar boom and the coming humira price war
-
 40:51
40:51
biologics and biosimilars interchangeability: a deep dive with industry experts
-
 4:20
4:20
the biosimilar development process
-
 57:41
57:41
webinar: biologics & biosimilars
-
 2:52
2:52
what's health canada saying about biosimilars?
-
 1:01
1:01
biosimilars video series: differences and similarities between biologics and biosimilars
-
 3:33
3:33
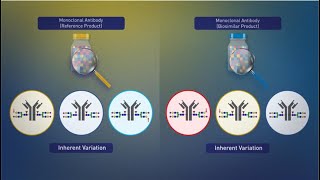
biosimilars: manufacturing and inherent variation