calculate the mass percent (m/m) of solution containing 26g of k2co3 and 724g of h2o
Published 3 months ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 31:25
31:25
molarity, molality, volume & mass percent, mole fraction & density - solution concentration problems
-
 4:30
4:30
calculate %m/m (percent by mass of a solution)
-
 8:12
8:12
how to calculate mass percent of solute and solvent of solution examples and practice problems
-
 0:33
0:33
what is the mass percent of solution containing 27.20 g sugar dissolved in 292.80 g water?
-
 3:29
3:29
calculate %m/v, mass-volume percent 2 examples
-
 5:51
5:51
how to solve percent concentration problems even if you're 🤷🏻♀️ - dr k
-
 9:08
9:08
college degree difficulty tier list (most difficult majors ranked)
-
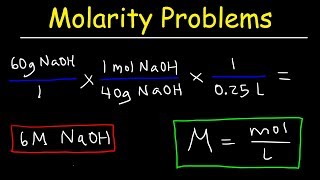 21:27
21:27
molarity practice problems
-
 3:23
3:23
calculate %v/v (percent by volume of a solution)
-
![calculate the [h3o ] of each aqueous solution with the following [oh- ]:](https://i.ytimg.com/vi/t7A3jkjCsf0/mqdefault.jpg) 0:33
0:33
calculate the [h3o ] of each aqueous solution with the following [oh- ]:
-
 0:29
0:29
which majors have the happiest students?
-
 0:33
0:33
what is the mass of solute dissolved in 15.0 ml of 6.00 m naoh?
-
 8:05
8:05
mass percent of a solution made easy: how to calculate mass % or make a specific concentration
-
 0:33
0:33
18. the vapor pressure of a solution containing nonvolatile solute is directly the proportional to …
-
 3:23
3:23
how to determine the amount of solution that contains a given amount of solute
-
 0:33
0:33
consider separate solutions of naoh and kcl made by dissolving 100.0 g of each solute in 250.0 ml o…
-
 0:33
0:33
make 100 ml of a solution that contains 0.5 mm mgcl2, 15 mm cacl2, and 120 mm kcl from 0.5 m stocks…
-
 0:33
0:33
a 1.461 g sample containing k2so4, nh4cl, and inert materials was dissolved in sufficient water to …
-
 0:33
0:33
what mass of potassium phosphate is needed for the preparation of a solution with a mass percent of…
-
 0:33
0:33
calculate the boiling points of each of the following solutions: a. 0.23 mm glucose in ethanol b. 2…
-
 0:33
0:33
what is the percent cscl by mass in a 0.711 m cscl solution that has density of 1.091 g/ml? selecte…
-
 0:33
0:33
what volume of a 0.700 m solution of k3po4 (formula mass = 212.27 g/mol) contains 53.1 g of the sol…