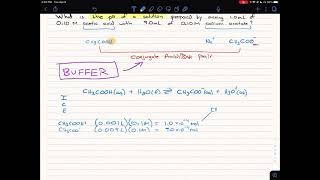
calculate the ph of a buffered solution of acetic acid and its acetate in 4 different concentrations
Published 4 years ago • 558 plays • Length 10:12Download video MP4
Download video MP3
Similar videos
-
 4:49
4:49
how to find the ph of a buffered solution containing acetic acid and sodium acetate - part 1
-
 33:21
33:21
buffer solutions
-
 4:43
4:43
find the ph of a buffer after adding hcl
-
 3:30
3:30
an acetic acid (pka = 4.74)/sodium acetate buffer with ph = 4.55 is needed for an experiment.
-
 3:29
3:29
how to find the ph of a buffered solution lactic acid and sodium lactate
-
 23:54
23:54
buffer solution, ph calculations, henderson hasselbalch equation, buffer solution ph calculations
-
 5:25
5:25
find the ph of a buffer after adding naoh
-
 28:17
28:17
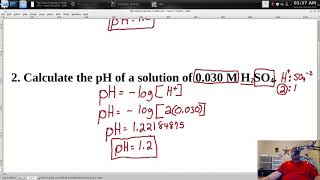
calculate the ph of acids and bases given the concentration of a solution
-
 11:39
11:39
buffer solution ph calculations | chemistry | khan academy
-
 7:05
7:05
calculate the ph of 0.1 m acetic acid
-
 7:19
7:19
calculate the ph of a buffer solution
-
 3:44
3:44
d.4 / b.7 calculating the ph of a buffer solution (sl / hl)
-
 5:14
5:14
calculating the ph of a buffer
-
 5:28
5:28
determining the ph of a buffer solution example calculation
-
 6:04
6:04
ph of buffer solution (example)
-
 6:38
6:38
aqueous solution equilibrium -02 calculating the ph of buffer solutions
-
 4:10
4:10
14.84b | what is the effect on the concentration of acetic acid, hydronium ion, and acetate ion
-
 3:23
3:23
calculate ph of acidic buffer
-
 6:46
6:46
⚗️ calculating the ph of a buffer solution (equilibrium method, question 1)
-
 13:27
13:27
buffer solution calculations 2
-
 7:32
7:32
the common-ion effect. calculate the ph of a solution containing a weak acid and its conjugate base.