calculate the ph of a solution that is 0.025 m in ch3cooh (ka = 1.8 x 10-5) and 0.025 m in hcn (ka …
Published 5 months ago • 2 plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 0:33
0:33
calculate the ph of a solution that is 0.15 m ch3cooh and 0.75 m ch3coona. (ka = 1.8 x 10-5)
-
 0:33
0:33
calculate the ph of a solution that is 0.250 m in sodium formate (hcoona) and 0.110 m in formic aci…
-
 0:33
0:33
calculate the ph of a solution that is 1.00 m hcn and 1.00 m hf. compare the conc…
-
 0:33
0:33
a buffer solution is prepared by taking 0.400 moles of acetic acid (pka 4.76) and 0.250 moles of ca…
-
 0:33
0:33
what is the ph of a solution prepared by mixing 30.00 ml of 0.10 m ch3co2h with 30.00 ml of 0.030 m…
-
 33:21
33:21
buffer solutions
-
 21:09
21:09
how to calculate the ph of a solution without a calculator - acids and bases
-
 28:17
28:17
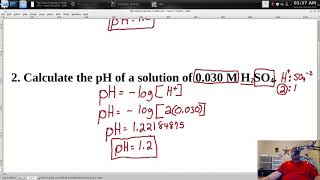
calculate the ph of acids and bases given the concentration of a solution
-
![the ph of a solution at 25 c in which [oh-] = 3.2 x 10^-5 m is ____. (kw= 1.01 x10^-14) a) 4.49 b) …](https://i.ytimg.com/vi/qrao2wy7i_I/mqdefault.jpg) 0:33
0:33
the ph of a solution at 25 c in which [oh-] = 3.2 x 10^-5 m is ____. (kw= 1.01 x10^-14) a) 4.49 b) …
-
 0:33
0:33
calculate the ph of 0.02 m nitric acid (hno3) solution. a buffer solution contains 2.9 m acetic aci…
-
 0:33
0:33
because of its functional group, a solution of ch3cooh dissolved in water could have a ph f 10 1*10
-
 0:33
0:33
in an experiment, a student titrated 50.0ml of 0.100m hc2h3o2 with 0.100m naoh(aq). calculate the p…
-
 0:33
0:33
for 270.0 ml of pure water, calculate the initial ph and the final ph after adding 0.032 mol of nao…
-
 0:33
0:33
a buffer solution is prepared by taking 0.250 moles of acetic acid ch3cooh (pka 4.76) and 400 moles…
-
 0:33
0:33
calculate the ph of a solution that is 0.125 m benzoic acid and 0.260 m sodium benzoate, a salt who…
-
 0:33
0:33
b. using a ph probe, the student determines that the ph of 0.115 m hc2h3o2(aq) is 2.92. using the p…
-
 0:33
0:33
calculate the ph in the solution formed by adding 10.0 ml of 0.050 m naoh to 40.0 ml of 0.0250 m be…
-
![what is the ph of a solution that contains [h ]=3.18 x 10^-8 m?](https://i.ytimg.com/vi/IJGLRfj0E0c/mqdefault.jpg) 0:33
0:33
what is the ph of a solution that contains [h ]=3.18 x 10^-8 m?
-
 0:33
0:33
question 11 (1 point) you wish to prepare a buffer of ph 9.90 by mixing hydrocyanic acid (hcn, aq) …
-
 0:33
0:33
if the initial concentration of an acid in solution is 2.30 m, and the solution has a ph = 3.40 at …
-
 0:33
0:33
a solution of ammonia has a ph of 11.8. what is the concentration of oh– ions in the solution? us…
-
![a buffer is prepared such that [ch3cooh]0 = [ch3coo-]0 = 0.050 m. calculate the volume of 0.10 m na…](https://i.ytimg.com/vi/wx4o19lRG3c/mqdefault.jpg) 0:33
0:33
a buffer is prepared such that [ch3cooh]0 = [ch3coo-]0 = 0.050 m. calculate the volume of 0.10 m na…