calculate the ph of each solution given the following [h3o ] or [oh-] values. [h3o ] = 9.0ã—10^-4 m…
Published 2 weeks ago • 4 plays • Length 0:33
Download video MP4
Download video MP3
Similar videos
-
 4:49
4:49
find the ka of an acid (given ph) (0.1 m hypochlorous acid) example
-
 0:33
0:33
calculate the ph of each of the following solutions: (a) 0.0010 m hcl, (b) 0.76 m…
-
 2:00
2:00
find the hydronium ion concentration given the ph
-
 0:33
0:33
a solution is 0.34 m nh3. calculate the ph of this solution? (kb = 1.8 x 10^–5)
-
 3:32
3:32
how to calculate the ph of a solution
-
 0:33
0:33
calculate the ph ofthe following solutions a 0.250 m solution of hf 7.1 x 10-') a 0.500 m solution …
-
 0:33
0:33
calculate the ph of each of the following solutions of a strong acid in water. a. 0.10 …
-
![ph calculations - calculate [h3o ] and [oh-], and find the ph of a solution](https://i.ytimg.com/vi/v67bWn-ogkE/mqdefault.jpg) 22:56
22:56
ph calculations - calculate [h3o ] and [oh-], and find the ph of a solution
-
 21:09
21:09
how to calculate the ph of a solution without a calculator - acids and bases
-
 5:49
5:49
pka, ka, and acid strength
-
 7:34
7:34
henderson–hasselbalch equation | acids and bases | ap chemistry | khan academy
-
 0:33
0:33
calculate the ph of each solution_ a solution containing 0.0314 m maleic acid and 0.045 m disodium …
-
 0:33
0:33
calculate the ph of each of the following solutions: (a) a sample of seawater that has an oh - conc…
-
 4:05
4:05
how to find ph, poh, h3o , and oh- step by step
-
 0:33
0:33
what is the ph of each of the following solutions? (a) 0.0045 m hcl ; (b) 6.14 …
-
 0:33
0:33
calculate the ph of each of the following solutions. a. 0.100 m propanoic acid (hc_3…
-
 0:33
0:33
calculate the ph values of the following solutions: a. a 1.0 m solution of acetic acid ≤…
-
 28:17
28:17
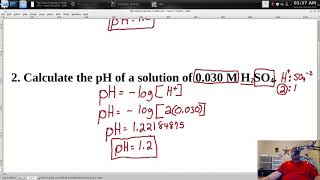
calculate the ph of acids and bases given the concentration of a solution
-
 0:33
0:33
a) calculate the concentrations of h , hco3-, and co3^2- in a 0.025 m h2co3 solution. b) calculate …
-
 0:33
0:33
calculate the hydrogen ion concentration and the ph of each of the following solutions of strong ac…
-
 0:33
0:33
calculate the ph of a solution containing 0.085 m nitrous acid (hno2, ka = 4.5 * 10-4) and 0.10 m p…
-
 0:33
0:33
the ka of propanoic acid (c2h5cooh) is 1.34ã—10^-5. calculate the ph of the solution and the concen…
Clip.africa.com - Privacy-policy
 4:49
4:49
 0:33
0:33
 2:00
2:00
 0:33
0:33
 3:32
3:32
 0:33
0:33
 0:33
0:33
![ph calculations - calculate [h3o ] and [oh-], and find the ph of a solution](https://i.ytimg.com/vi/v67bWn-ogkE/mqdefault.jpg) 22:56
22:56
 21:09
21:09
 5:49
5:49
 7:34
7:34
 0:33
0:33
 0:33
0:33
 4:05
4:05
 0:33
0:33
 0:33
0:33
 0:33
0:33
 28:17
28:17
 0:33
0:33
 0:33
0:33
 0:33
0:33
 0:33
0:33