calculate the uncertainty in the position `(delta x)` of an electron if `delta v is 0.1 %` .take...
Published 4 years ago • 1.9K plays • Length 2:27Download video MP4
Download video MP3
Similar videos
-
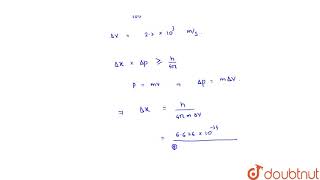 2:28
2:28
calculate the uncertainty in the position `(delta x)` of an electron if `delta v is 0.1 %`
-
 2:10
2:10
for an electron,if theuncertainty in velocity is delta vthe uncertainty in its position (delta ...
-
 6:23
6:23
calculate the uncertainty in position of an electron, if δv = 0.1% and υ = 2.2 x106ms-1
-
 2:25
2:25
for an electron if the uncertainty in velocity is \( \delta v \), t...
-
 17:18
17:18
heisenberg's uncertainty principle explained & simplified - position & momentum - chemistry problems
-
 3:52
3:52
suppose that the uncertainty in determining the position of an electron in an orbitis 0.6 å.
-
 6:28
6:28
quantum mechanics and the schrödinger equation
-
 5:13
5:13
heisenberg uncertainty principle example # 1
-
 5:55
5:55
⚗️ wavelength of light for a transition in the hydrogen atom (question 1)
-
 2:04
2:04
calculate the uncertainty in the velocity of an electron of the uncertainty in its position is
-
 0:24
0:24
isaac newton's insane sleep habits 😬
-
 1:41
1:41
an electron traveling at 1 35 105 ms has an uncertainty in its velocity of 1 88 105 ms what is th
-
 2:09
2:09
what is the uncertainty in velocity of an electron if the uncertainty in its position is
-
 5:05
5:05
the uncertainty in the position of an electron (mass `=9.1xx10^(-28) | class 12 chemistry | doubtnut
-
 2:18
2:18
if uncertainty in the position of an electron is zero, the uncertainty in its momentum would be....?
-
 2:13
2:13
the position of both, an electron and a helium atom is known within 1.0 nm. further the momentum
-
 5:54
5:54
how to compute the heisenberg uncertainty in position and momentum
-
 3:12
3:12
the uncertainty in momentum of an electron is1 × 10^–5 kg m/s. the uncertainty in its position
-
 6:13
6:13
uncertainty principle: numerical
-
 5:20
5:20
the heisenberg uncertainty principle part 2: energy/time and quantum fluctuation
-
 5:57
5:57
calculating the uncertainty in position of an electron
-
 2:08
2:08
what is the uncertainty in velocity of an electron if the uncertainty in its position is `10^(-1...