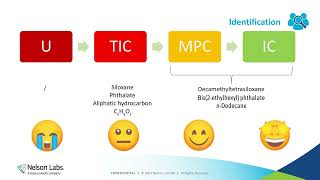
chemical characterization/toxicological risk assessments: a smart approach to biological evaluation
Published 4 years ago • 2.7K plays • Length 58:35Download video MP4
Download video MP3
Similar videos
-
 37:37
37:37
chemical characterization: how to initiate the biological evaluation of medical devices
-
 44:02
44:02
chemical characterization: the starting point for biological evaluations
-
 58:50
58:50
chemical characterization & toxicological risk assessment for medical device biocompatibility
-
 25:01
25:01
what are the four different types of medical device risk analysis?
-
 50:02
50:02
nitrosamines issue - a regulatory overview scientific webinar 2024
-
 1:05:08
1:05:08
understanding key components of a medical device clinical evaluation
-
 29:25
29:25
medical device chemical characterization
-
 1:08:16
1:08:16
biocompatibility testing of combination products & medical devices: new regulatory guidance
-
 31:13
31:13
biological evaluation of breathing gas pathways of medical devices, a new iso standard
-
 59:59
59:59
developing a biological safety evaluation
-
 1:01:28
1:01:28
develop a risk based biological safety evaluation per new us fda guidance: understand test options
-
 1:11:46
1:11:46
biological evaluation of medical devices webinar
-
 47:00
47:00
developing test strategies per the new iso 10993-1: current status and upcoming changes
-
 29:27
29:27
developing a testing plan for medical device design verification
-
 1:08:16
1:08:16
biocompatibility testing of combination products & medical devices: new regulatory guidance
-
 59:30
59:30
the testing and risk management impacts of changing a medical device
-
 1:42
1:42
medical device testing
-
 1:47
1:47
your trusted package testing partner
-
 1:01
1:01
eurofins biopharma product testing introduction
-
 32:42
32:42
how to perform a biological evaluation of your medical devices?