coa for rare diseases resource roll out
Published 8 months ago • 25 plays • Length 42:52Download video MP4
Download video MP3
Similar videos
-
 11:08
11:08
rare disease endpoint advancement rdea pilot program
-
 1:29:03
1:29:03
2021 pro consortium workshop: identifying coas for use in rare disease treatment trials
-
 4:17
4:17
rdca-dap: shortening the timeline for developing new treatments for rare diseases
-
 29:30
29:30
accelerating drug development for rare diseases through data sharing and analysis
-
 12:12
12:12
the 6 rare diseases changing modern medicine
-
 4:05
4:05
probability comparison: rarest mental disorders
-
 42:38
42:38
develop project schedule: the critical path method
-
 1:00:10
1:00:10
guids and de-identification tools for rare diseases
-
 2:33
2:33
rare disease patient voice: david fajgenbaum
-
 2:27:49
2:27:49
c-path rare & orphan disease conference pm sessions sept 11 2023
-
 46:37
46:37
clinical trial simulation tools advancing treatments in rare disease
-
 48:25
48:25
c-path rare & orphan disease conference day three am sessions sept 13, 2023
-
 22:01
22:01
optimization of clinical trial design for rare disease through modeling: example in duchenne’s
-
 2:04:49
2:04:49
rare diseases aggregated data bases to inform drug development research
-
 2:27
2:27
jane larkindale - why are c-path drug development tools important in rare disease drug development?
-
 57:10
57:10
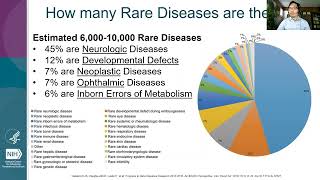
rare diseases are not rare – a training on rare disease resources
-
 31:11
31:11
from data to cures: speeding the way for rare diseases
-
 3:44
3:44
rare disease challenges in each step of the drug development process
-
 8:03
8:03
rdca-dap: how the data and analytics platform accelerates rare disease drug development
-
 3:27
3:27
what does the fda hope to achieve through developing the rare disease cures accelerator?