conducting your 1st internal audit for iso 13485:2016 certification
Published Streamed 3 years ago • 2.4K plays • Length 1:00:56Download video MP4
Download video MP3
Similar videos
-
 15:25
15:25
iso 13485: 2016 internal audit requirements l medical device internal audit l the learning reservoir
-
 1:24:32
1:24:32
six steps to iso 13485:2016 certification and mdsap certification
-
 6:35
6:35
internal audit for medical device manufacturers (iso 13485 compliance)
-
 0:41
0:41
online course iso 13485 internal audit medical devices
-
 1:52:50
1:52:50
md-qms full course of iso 13485:2016 | training on iso 13485:2016| training on full course |
-
 25:28
25:28
how to perform your internal audits correctly? (medical devices)
-
 1:30:47
1:30:47
mdsap medical device single audit program
-
 14:42
14:42
what is mdsap?
-
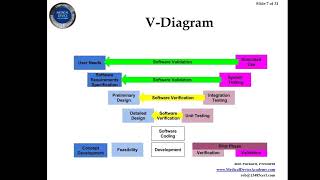 1:15:28
1:15:28
how to you create a design history file (dhf)?
-
 3:41
3:41
iso 13485:2016 internal auditor training kit | medical devices - quality management system
-
 1:40
1:40
licqual iso 13485 2016 medical devices quality management systems internal auditor
-
 2:03
2:03
online iso 13485:2016 lead auditor training
-
 11:12
11:12
what is iso 13485?
-
 2:37
2:37
what is iso 13485?
-
 6:22
6:22
internal auditing for iso 13485 (mdqms)
-
 4:58
4:58
iso 13485:2016 demo
-
 1:54:13
1:54:13
md-qms full course of iso 13485:2016 | training on iso 13485:2016| training on full course |
-
 5:57
5:57
iso 13485 internal auditor training | empowering assurance systems (eas)
-
 1:01
1:01
iso 13485 2016 training ppt presentation kit pa
-
 12:57
12:57
medical device quality management system md qms(iso13485:2016)
-
 20:59
20:59
how to write an iso 13485:2016 quality manual
-
 3:09
3:09
internal communication iso 13485 § 5.5.3 (executive series #62)