for the reaction: pcl5(g) ? pcl3(g) cl2(g); kp = 11.5 at 600.k suppose that 2.457 g pcl5 is place…
Published 2 weeks ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 0:33
0:33
for the reaction pcl5 (g) cl2 (g) = pci3 (g) at a particular temperature, kc = 24.3. suppose the …
-
 0:33
0:33
for the reaction pcl3(g) cl2(g) → pcl5(g) the initial mixture contains 4.35 mol of pcl3, 5.75 m…
-
 0:33
0:33
consider the following reversible reaction at equilibrium: co(g) cl2(g) cocl2(g). if chlorine gas…
-
 2:36
2:36
determining favorability of reactions
-
 0:33
0:33
find the ph of a buffer that consists of 0.50m methylamine (ch_3 nh_2…
-
 6:19
6:19
pcl-5 assessment
-
 12:18
12:18
【検査の簡単解説】sala失語症検査part2
-
 1:27:17
1:27:17
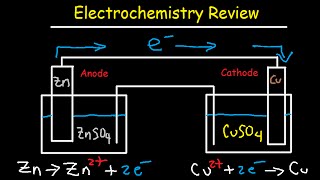
electrochemistry review - cell potential & notation, redox half reactions, nernst equation
-
 2:07:01
2:07:01
past year question sk025 2019/20 - explained!
-
 0:33
0:33
which of the following statements about ? decay are correct? (there may be more than one correct c…
-
 0:33
0:33
exercise: consider the following reaction: p (s) o2 (g) - p2o5 (s). if 6.25 g of phosphorus is b…
-
 18:38
18:38
rates of appearance, rates of disappearance and overall reaction rates
-
 6:33
6:33
finding ecell for a reaction
-
 0:33
0:33
write the mechanism of the reaction between p -hydroxyaniline and acetic anhydride to prepare aceta…
-
 10:58
10:58
chapter 15 – chemical equilibrium: part 5 of 12
-
 54:00
54:00
ap chemistry 5.1-5.3 reaction rates, rate law, and concentration changes
-
 3:03
3:03
for the reaction n2o5(g)→2no2(g) 1/2o2(g) the value of rate of disappearance of n2o5 is given as
-
 0:33
0:33
post-lab questions: 1. can you dissolve 0.35 moles of potassium permanganate (kmno4) into 500 ml of…
-
 0:33
0:33
determine the number of grams of hcl that can react with 0.750 grams of al(oh)3 according to the fo…
-
 22:31
22:31
ch17e 5 more on consecutive elementary reactions
-
 0:33
0:33
from the following rate constants, determined at five temperatures, calculate the experimental ener…