consider the following chemical reaction at equilibrium: 2no2(g) ⇜ n2o4(g). if the container's vo…
Published 3 days ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 44:45
44:45
gibbs free energy - entropy, enthalpy & equilibrium constant k
-
 4:08
4:08
consider the reaction, n_2(g) 3 h_2(g) ⇌ 2 nh_3(g) the equilibrium constant of the above re...
-
 2:54
2:54
13.86a | consider the equilibrium 4no2(g) 6h2o(g) ⇌ 4nh3(g) 7o2(g) (a) what is the expression
-
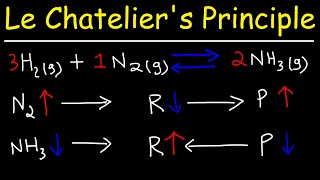 26:40
26:40
le chatelier's principle
-
 9:13
9:13
16.45 | consider the following reaction at 298 k: n2o4(g) ⇌ 2no2(g) kp = 0.142 what is the standard
-
 0:33
0:33
for the reaction, h2(g) i2(g) ⇜ 2hi(g) kc = 55.3 at 700 k. in a 2.00-l flask containing an equi…
-
 53:22
53:22
chemical equilibrium constant k - ice tables - kp and kc
-
 1:16
1:16
consider the reaction 2nh3(g) → n2(g) 3h2(g)
-
 1:06:01
1:06:01
chemical kinetics practice problems - complete review
-
 34:53
34:53
chemical kinetics - initial rates method
-
 38:21
38:21
15. thermodynamics: bond and reaction enthalpies
-
 0:50
0:50
for the reaction h2(g) i2(g)⇌2hi(g), the rate of reaction is expressed as
-
 0:33
0:33
consider the reaction: co(g) h2o(g) ⇜ co2(g) h2(g) k = 130 at 500 k if a reaction mixture ini…
-
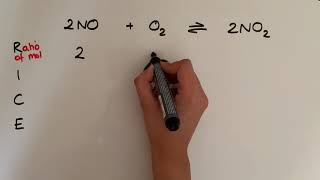 10:13
10:13
using rice to calculate equilibrium concentrations
-
 10:51
10:51
how to calculate kp from kc - chemical equilibrium
-
 4:27
4:27
chemical equilibrium - formation of hydrogen iodide
-
 3:02
3:02
practice problem: calculating equilibrium concentrations
-
 0:58
0:58
calculate base dissociation constant of conjugate base from ionic product of water @chemistryguru
-
 0:46
0:46
chemical equilibrium iit&jee questions no 11 || ix class
-
 7:32
7:32
at 700k, equilibrium constant for the reaction: h2 (g) i2 (g) ⇌ 2hi (g)is 54.8...
-
 4:26
4:26
understanding free energy and equilibrium | professor dave & chegg explain
-
 1:16
1:16
consider the reaction : n2(g) 3h2(g)→2nh3(g)