design history file 820.30j & iso 13485 § 7.3.10 (executive series #19)
Published 3 years ago • 4.2K plays • Length 3:29Download video MP4
Download video MP3
Similar videos
-
 3:22
3:22
design inputs 820.30c & iso 13485 § 7.3.3 (executive series #12)
-
 3:51
3:51
design review 820.30e & iso 13485 § 7.3.5 (executive series #14)
-
 3:32
3:32
design outputs 820.30d & iso 13485 § 7.3.4 (executive series #13)
-
 3:29
3:29
design changes 820.30i & iso 13485 § 7.3.9 (executive series #18)
-
 3:44
3:44
design controls 820.30 & iso 13485 § 7.3 (executive series #9)
-
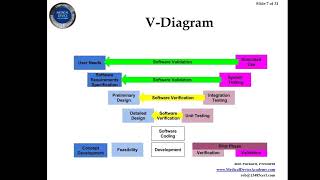 1:15:28
1:15:28
how to you create a design history file (dhf)?
-
 3:47
3:47
design validation 820.30g & iso 13485 § 7.3.7 (executive series #16)
-
 15:40
15:40
format & content of a medical device file (mdf) - iso 13485:2016, clause 4.2.3
-
 1:24:32
1:24:32
six steps to iso 13485:2016 certification and mdsap certification
-
 20:59
20:59
how to write an iso 13485:2016 quality manual
-
 3:17
3:17
design transfer 820.30h & iso 13485 § 7.3.8 (executive series #17)
-
 2:31
2:31
design history file consultant - operon strategist
-
 3:29
3:29
device history record 820.184 & iso 13485 § 7.5.1 (executive series #25)
-
 3:29
3:29
design verification 820.30f & iso 13485 § 7.3.6 (executive series #15)
-
 3:09
3:09
internal communication iso 13485 § 5.5.3 (executive series #62)
-
 3:45
3:45
device master record 820.181 & iso 13485 § 4.2.3 medical device file (executive series #24)
-
 12:48
12:48
why does 21 cfr 820 need to be modernized to iso 13485?
-
 3:24
3:24
process changes 820.70b and iso 13485 § 4.1.4, 4.2.4, 7.3.9, 7.4.3, 7.5.6 (executive series #32)
-
 11:47
11:47
revolutionize compliance: shifting from fda 21 cfr part 820 to iso 13485
-
 19:07
19:07
fda's transition from cfr 820 to the iso 13485:2016 instituting a new qms
-
 3:19
3:19
buildings 820.70f & iso 13485 § 6.3, 7.5.1. (executive series #36)