design history file dhf, device master record dmr, device history record dhr and technical file tf
Published 8 years ago • 14K plays • Length 1:02:57Download video MP4
Download video MP3
Similar videos
-
 11:01
11:01
design history file (dhf), the device master record (dmr) and the device history record (dhr)
-
 12:00
12:00
design history file (dhf), the device master record (dmr) and the device history record (dhr)
-
 3:45
3:45
device master record 820.181 & iso 13485 § 4.2.3 medical device file (executive series #24)
-
 11:29
11:29
key regulatory documents design history file dhf, device master record dmr, device history record d
-
 4:46
4:46
device history record vs. device master record l 21 cfr 820 dhr dmr l the learning reservoir
-
 48:16
48:16
preparing a device master record (dmr)
-
 1:53
1:53
design history file - 77 second webinar #23
-
 1:15:28
1:15:28
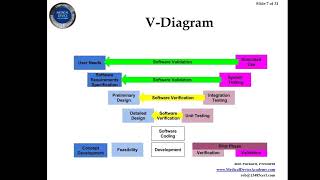
how to you create a design history file (dhf)?
-
 48:16
48:16
preparing a device master record (dmr)
-
 18:40
18:40
hl7 fhir cds hooks - a practical guide
-
 59:53
59:53
medical device design control
-
 1:09:32
1:09:32
dhf, dmr, dhr and tf regulatory documents explained
-
 1:09:32
1:09:32
regulatory documents explained - dhf, dmr, dhr and tf
-
 59:20
59:20
device master record & device history record - a regulatory
-
 14:07
14:07
dhf, dmr, dhr, technical file and design dossier - key requirements and future directions
-
 0:55
0:55
dhf/ dhr/ dmr
-
 4:53
4:53
design history file, device history record, device master record and impact of change controls
-
 47:38
47:38
is it time to re-evaluate your design history file (dhf)?
-
 2:40
2:40
dmr and dhr fda compliance made easy - erp & qms for medical devices
-
 58:34
58:34
documentation deconstructed: understanding the technical file
-
 3:29
3:29
device history record 820.184 & iso 13485 § 7.5.1 (executive series #25)