determine the ph of each of the following solutions: a) a solution that is 4.2ã—10^(-2) m in hclo4 …
Published 4 months ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 0:33
0:33
calculate the ph of a 0.30 m naf solution. (ka for hf = 7.2ã—10^-4)
-
![chm 1032 in-class quiz #5: chapters 9 what is the ph of a solution that has a [h3o ] = 3.4 x 10^27?…](https://i.ytimg.com/vi/iIo1esFJcUQ/mqdefault.jpg) 0:33
0:33
chm 1032 in-class quiz #5: chapters 9 what is the ph of a solution that has a [h3o ] = 3.4 x 10^27?…
-
 21:09
21:09
how to calculate the ph of a solution without a calculator - acids and bases
-
 0:33
0:33
calculate the ph of the following solutions (without considering the activities). a) pure water at …
-
 0:33
0:33
1. calculate the ph of 0.00200 m solution of perchloric acid, hclo4. 2. calculate the ph of 0.00345…
-
 0:33
0:33
use the henderson - hasselbalch equation to calculate the ph of each solutiona. a solution that is …
-
 28:17
28:17
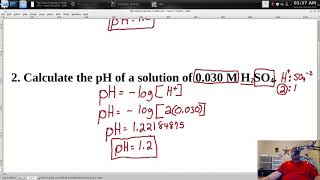
calculate the ph of acids and bases given the concentration of a solution
-
 3:14
3:14
how to calculate concentration from ph and poh
-
 5:04
5:04
acid-base equilibria and buffer solutions
-
 0:33
0:33
calculate the ph of each of the following solutions: (a) 0.0010 m hcl, (b) 0.76 m…
-
 0:33
0:33
calculate the ph of a solution that is 0.250 m in sodium formate (hcoona) and 0.110 m in formic aci…
-
 0:33
0:33
what is the ph of 4 , solution that results from mixing 25.0 ml of 0.200 m ha with 12.5 ml of 2-30.…
-
 33:21
33:21
buffer solutions
-
 0:33
0:33
what is the ph of a solution prepared by mixing 30.00 ml of 0.10 m ch3co2h with 30.00 ml of 0.030 m…
-
 0:33
0:33
what is the ph of each of the following solutions? (a) 0.0045 m hcl ; (b) 6.14 …
-
![ph calculations - calculate [h3o ] and [oh-], and find the ph of a solution](https://i.ytimg.com/vi/v67bWn-ogkE/mqdefault.jpg) 22:56
22:56
ph calculations - calculate [h3o ] and [oh-], and find the ph of a solution
-
 7:04
7:04
14.19a | calculate the ph and the poh of 0.000259 m hclo4
-
 0:33
0:33
calculate the hydrogen ion concentration for an aqueous solution that has a ph of 3.45. 1. 0.54 m 2…
-
 0:33
0:33
4.what is the ph of a 6.50 x 10-3 m koh solution? (hint: this is a basic solution – concentration i…
-
 0:33
0:33
calculate the ph of a solution prepared by dissolving 0.050 mol of benzoic acid (ha) and 0.50 mol o…
-
 0:33
0:33
calculate the ph at the following points in the titration of 50 ml of 0.01 m naoh with 0.1 m hcl. (…
-
 0:33
0:33
calculate the concentration of pb^2 in each of the following. a. a saturated solution o…