gore c3 excluder aaa endoprosthesis deployment
Published 5 months ago • 570 plays • Length 1:32Download video MP4
Download video MP3
Similar videos
-
 4:54
4:54
understanding the mdcg 2021-24: navigating the complex world of medical device classification
-
 8:08
8:08
37 basic medical equipments with names and their uses
-
 4:08
4:08
bd facsdiva™ software part 3, area scaling
-
 0:17
0:17
dermoscopy equipment case large del l 117
-
 0:36
0:36
guardant360® cdx receives fda approval
-
 21:16
21:16
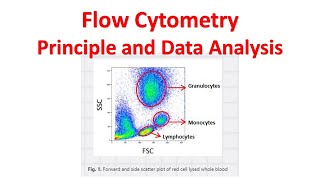
flow cytometry tutorial | flow cytometry data analysis | flow cytometry gating
-
 3:21
3:21
transfer an exoplan implant plan to exocad and 3d print crown in 10 mins! - idda minibytes tutorials
-
 11:12
11:12
what is iso 13485?
-
 1:50
1:50
brazil medical device registration process chapter 3 - registration holder
-
 2:22
2:22
fluid balance. diaverum d.academy
-
 1:04
1:04
meet obgyn linda fan, md, facog
-
 8:28
8:28
what is iso 13485 for medical devices?
-
 7:25
7:25
advanced medical directives in malaysia & indonesia / mjsl aug. 2024 (vol.12, issue 2)
-
 3:25
3:25
united states medical device registration chapter 3 - quality management system
-
 2:12
2:12
implant 3d tutorial - import dicom dicomdir
-
 0:55
0:55
eudamed registration: register your medical device or ivd for eu compliance
-
 0:26
0:26
why choose gsp customized color kinesiology tape? - waterproof kinesio tape
-
 1:32
1:32
south korea medical device registration chapter 3 - key players
-
 9:10
9:10
fda approvals in urothelial cancer and gist, promising data in urothelial carcinoma, and more
-
 2:16
2:16
china medical device and ivd classification
-
 4:10
4:10
5 useful resources when writing a medical device clinical evaluation report (cer)
-
 2:33
2:33
muller fabbri, md, phd