elementary rate laws - unimolecular, bimolecular and termolecular reactions - chemical kinetics
Published 6 years ago • 130K plays • Length 4:28Download video MP4
Download video MP3
Similar videos
-
 18:48
18:48
writing rate laws of reaction mechanisms using the rate determining step - chemical kinetics
-
 1:55
1:55
how to determine molecularity of a reaction examples (unimolecular, bimolecular, termolecular)
-
 48:46
48:46
integrated rate laws - zero, first, & second order reactions - chemical kinetics
-
 34:53
34:53
chemical kinetics - initial rates method
-
 9:10
9:10
kinetics: initial rates and integrated rate laws
-
 36:56
36:56
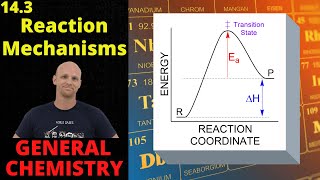
14.3 reaction mechanisms, catalysts, and reaction coordinate diagrams | general chemistry
-
 1:06:01
1:06:01
chemical kinetics practice problems - complete review
-
 1:04:19
1:04:19
chemical kinetics full review
-
 42:49
42:49
chemical kinetics summary in 30 minutes
-
 6:14
6:14
factors affecting the rate of the reaction - chemical kinetics
-
 7:31
7:31
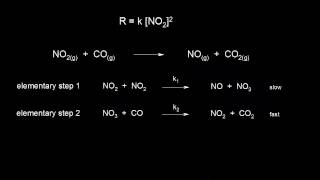
elementary reactions
-
 8:42
8:42
reaction mechanism and rate law | kinetics | ap chemistry | khan academy
-
 6:13
6:13
elementary reactions
-
 5:15
5:15
how to identify the intermediate & catalyst in a reaction mechanism - kinetics chemistry
-
 12:15
12:15
how to determine the units of the rate constant k - chemical kinetics
-
 5:35
5:35
elementary reactions | kinetics | ap chemistry | khan academy
-
 14:16
14:16
chapter 14 – chemical kinetics: part 5 of 17
-
 10:14
10:14
elementary rate laws
-
 10:01
10:01
r2.2.9 reaction mechanisms (hl)
-
 31:50
31:50
collision theory - arrhenius equation & activation energy - chemical kinetics