fda approval of a biosimilar, priority review in epithelioid sarcoma, and more
Published 4 years ago • 225 plays • Length 8:21Download video MP4
Download video MP3
Similar videos
-
 9:02
9:02
fda approval in sarcoma, priority reviews in mcrpc and myeloma, and more
-
 9:54
9:54
fda approval in nsclc, priority review designation in sclc, crl to biosimilar, and more
-
 10:01
10:01
fda approvals in sclc and a biosimilar, and japanese approvals in aml, ntrk tumors, and more
-
 5:04
5:04
epithelioid sarcoma: 5-minute pathology pearls
-
 1:55
1:55
dr. hurvitz on the fda approval process for biosimilars
-
 8:31
8:31
fda d.i.s.c.o.: first biosimilar approval for the treatment of cancer
-
 5:41
5:41
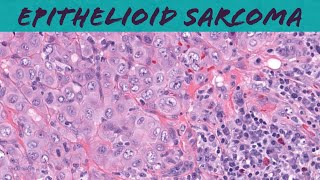
epithelioid sarcoma (explained in 5 minutes) - pathology dermpath dermatology dermatopathology
-
 6:18
6:18
epithelioid sarcoma explained in 5 minutes (pathology dermpath dermatopathology dermatology)
-
 1:17
1:17
dr. ferris on fda approval of nivolumab in patients with head and neck cancer
-
 1:55
1:55
dr. federman on use of nanotechnology in sarcomas
-
 5:57
5:57
evofosfamide versus ifosfamide patient selection in soft tissue sarcoma
-
 6:22
6:22
a voice for epithelioid sarcoma – nick saleum’s cancer journey
-
 8:57
8:57
epithelioid leiomyosarcoma
-
 2:59
2:59
idim: fungiscope - call for ifis due to paecilomyces or purpureocillium spp.
-
 20:20
20:20
regulatory trials in tumors
-
 0:53
0:53
dr. puhalla on the value of biosimilars in oncology
-
 2:02
2:02
hidradenoma papilliferum - histopathology
-
 7:24
7:24
epithelioid sarcoma - pathology mini tutorials
-
 1:48
1:48
papillary endothelial hyperplasia (masson tumor) - histopathology