fda guidance: definitions: applying human factors and usability engineering to medical devices
Published 4 years ago • 92 plays • Length 3:50Download video MP4
Download video MP3
Similar videos
-
 6:04
6:04
fda guidance: overview: applying human factors and usability engineering to medical devices
-
 6:31
6:31
introduction: fda guidance: applying human factors and usability engineering to medical devices
-
 18:29
18:29
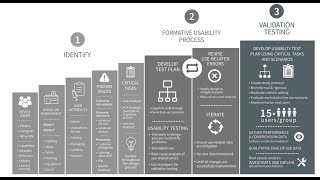
fda human factors guidance simplified
-
 50:57
50:57
the global guide to human factors and usability engineering regulations
-
 10:26
10:26
fda guidance: device users, use environments and user interface section 5
-
 8:51
8:51
human factors and usability testing for a 510(k) submission
-
 1:00:11
1:00:11
human factors engineering in medical products reviews (2of14) redi 2018
-
 11:50
11:50
what is the best way to perform risk management?
-
 12:15
12:15
510(k) tips and tricks
-
 12:26
12:26
introduction to human factors for investigations
-
 21:59
21:59
fda guidance: preliminary analyses and evaluations (section 6)
-
 1:27:09
1:27:09
webinar: applying human factors and usability engineering to medical devices
-
 4:00
4:00
human factors - fda definitions
-
 16:52
16:52
what’s the difference between fda human factors requirements and iec 62366?
-
 13:09
13:09
human factors process, can we make this easy to understand?
-
 1:10:44
1:10:44
demystifying fda's human factors guidance
-
 0:45
0:45
usability engineering fda process flow diagram human factors engineering tip for april 8th 2024
-
 0:59
0:59
example of human factors testing: simulated use test
-
 0:57
0:57
verify the fda's extent of recognition before utilizing consensus standards #fda #medicaldevices
-
 36:26
36:26
why do human factors usability matter for medical devices?