given the following data: 2 o_3(g) →3 o_2(g) …
Published 7 days ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 0:33
0:33
given the following data: 2o3(g) → 3o2(g) î”h = -427 kj o2(g) → 2o(g) î”h = 495 kj no(g) o3(g…
-
 0:33
0:33
given the following data: s(s) 3/2 o_2(g) →s o_3(g) δh=-395.2 …
-
 0:33
0:33
given the thermal chemical equation 2so2(g) o2(g) → 2so3(g) (î”h = -180.2 kj/mol), calculate th…
-
 2:42
2:42
calculate δh for the process: n2(g) 2o2(g) ⟶ 2no2(g)
-
 0:33
0:33
consider the reaction: 2clf(g) 2nh3(g) → 3hf(g) n2(g) cl2(g) î”hâ° = -1196 kj given the fol…
-
 0:33
0:33
(q.3: 5 points) you are given the following data for the decomposition of acetaldehyde (ch3cho): ch…
-
 0:33
0:33
calculate the enthalpy of the following chemical reaction: cs2(l) 3o2(g) → co2(l) 2so2(g) giv…
-
 4:22
4:22
arrhenius plot
-
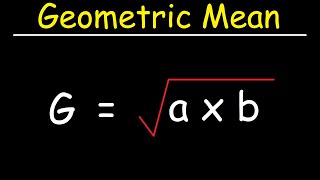 5:56
5:56
how to calculate the geometric mean
-
 14:03
14:03
hess's law problems & enthalpy change - chemistry
-
 0:33
0:33
for the reaction, 2 a(g) 2 b(g) = c(g) 3 d(g), the following data were obtained at constant te…
-
 0:33
0:33
given the following reactions: az (g) dz (g) → 2ad (g) y2 az (g) dz (g) → 7 adz (g) î”hrxn …
-
 0:33
0:33
consider the following reaction: 2 h2s (g) 3 o2 (g) → 2 so2 (g) 2 h2o (g) calculate the stand…
-
 0:33
0:33
calculate the reaction rate given the following: after 2 minutes (120 seconds), the concentration o…
-
 0:33
0:33
from the following data, calculate the average bond enthalpy for the n-h bond: …̱
-
 3:57
3:57
calculating enthalpy of reaction
-
 0:33
0:33
given the following set of data: mass of hydrated salt = 1.14 g mass of water = 0.50 g molar mass o…
-
 0:33
0:33
calculate the selectivity for propyl paraben compared to methyl paraben given the following data: t…
-
 1:45
1:45
given n_2( g) 3 h_2( g) → 2 nh_3( g) ; δ_r h^⊖=-92.4 kj mol^-1 what is the standard enthalpy of ...
-
 0:33
0:33
question: given the data below, how many grams of co2 would you expect to be formed in the reaction…
-
 0:33
0:33
consider the reaction 2 n2o5(g) d 4 no2(g) o2(g) a system at equilibrium has the following concen…
-
 0:33
0:33
the equilibrium between ozone and dioxygen is represented by: 2o3 (g) ⇜ 3o2 (g) kc = 2.5 ã— 10^15…