greenlight guru clinical | edc software overview
Published 6 months ago • 191 plays • Length 4:08Download video MP4
Download video MP3
Similar videos
-
 3:21
3:21
qms software for medical devices | greenlight guru product overview
-
 50:04
50:04
how electronic data capture is transforming the medtech industry
-
 15:14
15:14
greenlight guru academy medical device hq team up
-
 37:27
37:27
determining user needs for your medical device
-
 1:30:26
1:30:26
understanding the medical device classification system
-
 1:18:37
1:18:37
how to use clinical data for medical device submissions in both eu & us
-
 1:06:05
1:06:05
verification & testing strategies for compliance with iso 13485:2016 & iec 62304, 60601-1, 82304-1
-
![[global medical device podcast live] design controls, development, and risk for samd](https://i.ytimg.com/vi/AQ-Sv97X7L8/mqdefault.jpg) 40:09
40:09
[global medical device podcast live] design controls, development, and risk for samd
-
 56:15
56:15
key components of an effective post-market surveillance system
-
 2:31
2:31
photonicare adopts medical device qms software to accelerate time to market
-
 3:06
3:06
bioclinica edc demo video 2021
-
 45:54
45:54
key considerations for medical device cloud connectivity
-
![medical device design control: waterfall or agile? [jon speer greenlight guru]](https://i.ytimg.com/vi/-WqOtzkiM5E/mqdefault.jpg) 47:37
47:37
medical device design control: waterfall or agile? [jon speer greenlight guru]
-
 1:00:41
1:00:41
strategies for improving medical device software and cybersecurity
-
 47:09
47:09
software as a medical device (samd) challenges and opportunities for 2021 and beyond
-
 24:00
24:00
managing clinical data activities
-
 33:17
33:17
what is the safer technologies program (step) and can your device use it?
-
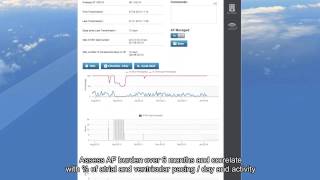 2:00
2:00
afinder demo
-
 50:28
50:28
why design validation is more than testing: how to validate your validation
-
 25:04
25:04
augmented reality (ar) in healthcare