how many ml hcl of a 0.100 m hcl solution is needed to completely neutralize 25.0 ml of a 0.350 m n…
Published 7 days ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 5:33
5:33
how many ml of 0.160 m hclo4 solution are needed to neutralize 33.5 ml of 0.0880 m naoh?
-
 0:33
0:33
calculate the volume of 0.100 m hcl solution needed to neutralize 25.0 ml of 0.350 m naoh solution.…
-
 0:33
0:33
if 18.0 ml of a 0.150 m naoh solution is required to neutralize 25.0 ml of an hcl solution, what is…
-
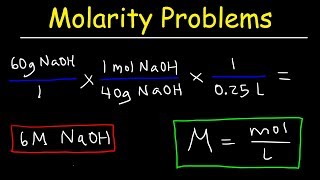 21:27
21:27
molarity practice problems
-
 1:33
1:33
sodium hydroxide (naoh) and hydrochloric acid (hcl) reaction l amazing science experiment
-
 31:25
31:25
molarity, molality, volume & mass percent, mole fraction & density - solution concentration problems
-
 0:33
0:33
(a) how many milliliters of 0.120 m hcl are needed to completely neutralize 50.0 m…
-
 4:56
4:56
1 gram of sodium hydroxide was treated with 25 ml of 0.75 m hcl solution, the mass of naoh left?
-
 4:51
4:51
if 12.5 ml of a 0.144 m naoh solution is required for neutralization of 6.0 ml of given hcl solution
-
 3:38
3:38
a hydrochloric acid solution will neutralize a sodium hydroxide solution consider these molecular v
-
 0:06
0:06
quiz 253 : if 0.5 n hcl is required to neutralize 0.25 n naoh, what is the ratio of their volumes?
-
 0:33
0:33
assigned as homework 0 q16.39 a 10.0 ml sample of an hcl solution is titrated with 1.00 m naoh solu…
-
 3:01
3:01
20.0 ml of a 3.0m hcl solution are mixed with 20.0 ml of a 5.0m naoh solution. what is the ph?
-
 0:33
0:33
if 10.0 ml of 0.100 m hcl is titrated with 0.200 m naoh, what volume of sodium hydroxide solution i…
-
 4:33
4:33
how many ml of 1m h2so4 is needed to neutralize 10ml of 1m of mg(oh)2?
-
 2:15
2:15
how to make a 0.1m hcl solution (hydrochloric acid)
-
 2:05
2:05
oqv no – 285 what volume of 0.4 (m) naoh is needed to neutralize 50 ml of 0.2 (m) hcl?
-
 0:20
0:20
chemistry || experiment no. : 08 || 12th standard || answers
-
 0:41
0:41
experiment to show #turmeric (#haldi ) as a natural #indicator..! #red #colour in #detergent (base)
-
 11:04
11:04
100. ml of 0.200 m hcl is titrated with 0.250 m naoh. part a 1)what is
-
 0:33
0:33
50.0 ml of a 3.00 m hcl solution is diluted to a total volume of 217.4 ml. what is the concentratio…
-
 1:50
1:50
10 ml of a solution of naoh is found to be completely neutralised by 8 ml of a given cbse class 10