how to structure a risk statement in risk-based monitoring for clinical trials?
Published 8 years ago • 234 plays • Length 2:10Download video MP4
Download video MP3
Similar videos
-
 6:00
6:00
risk-based monitoring guidelines
-
 18:18
18:18
risk-based monitoring - risk-optimized approaches to clinical trials - introduction - part 1 of 3
-
 2:11
2:11
risk assessment for risk-based monitoring
-
 6:03
6:03
risk based monitoring plan
-
 7:31
7:31
cra basics: what is risk-based monitoring in clinical research?
-
 3:02
3:02
risk documentation
-
 2:10
2:10
how do you decide what actions to take in risk-based monitoring clinical trials?
-
 52:37
52:37
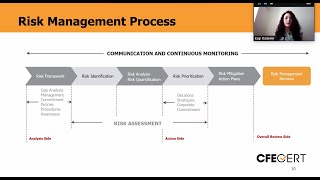
enterprise risk management - erm
-
 30:31
30:31
nist risk management frame (rmf)/categorize information system - rmf step 2
-
 5:29
5:29
risk and how to use a risk matrix
-
 4:36
4:36
risk based monitoring plan - what should not be done?
-
 13:41
13:41
risk-based monitoring - risk-optimized approaches to clinical trials - part 2 of 3
-
 1:09:46
1:09:46
risk-based monitoring for clinical research leaders
-
 2:11
2:11
background to risk-based monitoring clinical trials
-
 6:32
6:32
risk-based monitoring methods, strategy, and pitfalls
-
 4:52
4:52
risk breakdown structure
-
 55:31
55:31
quality by design in clinical trials lessons learned in implementing risk based monitoring
-
 6:46
6:46
managing the risks of clinical research projects with the scto's risk assessment form
-
 19:38
19:38
quality by design in clinical trials risk assessment (part 1)
-
 6:47
6:47
risk-based monitoring and quality by design in clinical trials