if it takes 50 ml of .5 m ca(oh) solution to completely neutralize 125 ml of hno3 solution, what is…
Published 4 days ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 0:33
0:33
calculate the volume (ml) of 0.100 m h3po4 required to neutralize 25.0 ml of 0.010 m ca(oh)2.
-
 5:33
5:33
how many ml of 0.160 m hclo4 solution are needed to neutralize 33.5 ml of 0.0880 m naoh?
-
 0:33
0:33
a solution of 170 ml of 5.5 m koh is added to 140 ml of 3.8 m ca(oh)2. calculate the total hydroxid…
-
 0:33
0:33
- in a titration, 27.4 ml of 0.0154 m ca(oh)2 is added to a 20 ml sample of hcl solution of unknown…
-
 14:00
14:00
#calculate the ph of 10-³ m hcl & 0.001m naoh solution
-
 0:33
0:33
we need to neutralize 200.0 ml of a 0.1 m solution of ca(oh)2 before we can dispose of it safely. h…
-
 17:36
17:36
dilution and concentration calculations in pharmacy | 5 key examples solved
-
 18:49
18:49
preparing solutions - part 3: dilutions from stock solutions
-
 21:55
21:55
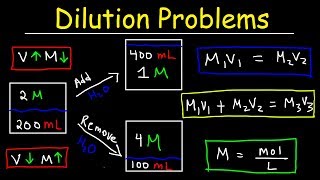
dilution problems, chemistry, molarity & concentration examples, formula & equations
-
 2:14
2:14
calculate the normality of 0.74 g ca(oh)_(2) in 10 ml of solution. | 11 | mole and equivalent co...
-
 2:14
2:14
calculate the normality of `0.74 g ca(oh)_(2)` in `10 ml` of solution.
-
 4:23
4:23
600 ml of `hno_3` and 200 ml of `ca(oh)_2` of same molarity are mixed to give a resulting soluti...
-
 4:20
4:20
a quantity of 19.50 ml of a koh solution is needed to neutralize 0.4000 g of khp.
-
 7:03
7:03
how solve dilution and concentration calculation problems 4
-
 7:29
7:29
how solve dilution and concentration calculation problems 5
-
 5:07
5:07
calculate the concentration (in molarity) of a naoh solution if 26.0 ml of the solution are needed
-
 0:33
0:33
calculate the volume of 0.125m hno3 required to neutralize 25.0ml of 0.250 m naoh
-
 0:33
0:33
how many grams of water are produced if 27.4 ml of the 0.164 m calcium hydroxide solution is reacte…
-
 0:33
0:33
on performing titration of 20 ml of ca(oh)2 solution of unknown concentration with 0.1 n h2s04, 25 …
-
 0:33
0:33
calculate the ph of the resulting dissolution from adding a) 0 ml b) 5 ml c) 7.5 ml d) 15 ml and e)…
-
 2:19
2:19
titration q1 (method 1)