if the rate of reaction (20.3) is 5.7 ×10^-4 m s^-1, what is the rate o…
Published 4 weeks ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 9:10
9:10
kinetics: initial rates and integrated rate laws
-
 0:33
0:33
if the rate of the reverse reaction is much slower than the rate of the forward reaction, does the …
-
 0:33
0:33
what is meant by the rate of a chemical reaction? what are the units of the rate of a reaction?
-
 0:33
0:33
if the rate of the reverse reaction is much slower than the rate of the forward reaction, does the …
-
 10:49
10:49
solving a rate law using the initial rates method
-
 0:33
0:33
consider the following reaction: ch_3-cl i^- ⟶…
-
 0:33
0:33
what is the change in the rate of the forward reaction after the reaction is at equilibrium?
-
 18:48
18:48
writing rate laws of reaction mechanisms using the rate determining step - chemical kinetics
-
 48:46
48:46
integrated rate laws - zero, first, & second order reactions - chemical kinetics
-
 31:01
31:01
first order and second order chemical kinetics example problems
-
 8:44
8:44
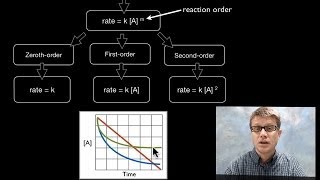
the rate law
-
 0:19
0:19
a satisfying chemical reaction
-
 16:26
16:26
chem 102 chapter 12-1 reaction rates
-
 11:22
11:22
chem 102 chapter 12-4 pseudo rate laws
-
 0:33
0:33
(a) a certain first-order reaction has a rate constant of 2.75 ×10^-2 s^-1 at 20…
-
 12:21
12:21
chem 102 chapter 12-2 rate laws
-
 30:45
30:45
rate law intro
-
 0:33
0:33
what is the average rate of the reaction in the first 22.3 s expressed in moles of phenolphthalein …
-
 0:58
0:58
ap chemistry kinetics rate- determining step practice problem
-
 14:26
14:26
chem 102 chapter 12-6 the arrhenius equation
-
 0:33
0:33
dinitrogen pentoxide decomposes in chloroform at a rate of 2.48 ×10^-4 mol /(…
-
 0:33
0:33
the order of a reaction is independent of temperature, but the value of the rate constant varies wi…