in atoms there is a finite, though very small, probability that, at some instant, an orbital electr…
Published 2 months ago • No plays • Length 0:33Download video MP4
Download video MP3
Similar videos
-
 0:33
0:33
(a) what is the probability that an electron in the 1s state of a hydrogen atom will be found at a …
-
 0:48
0:48
how small are atoms?
-
 5:36
5:36
examining a radial probability distribution to identify atomic orbitals su 2 b7
-
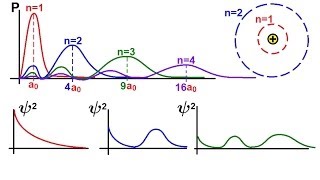 7:14
7:14
chemistry - electron structures in atoms (26 of 40) radial probability density function: s-orbital
-
 9:17
9:17
what if the universe is an atom? | unveiled
-
 5:23
5:23
where do electrons get energy to spin around an atom's nucleus?
-
 21:59
21:59
why don't electrons fall onto the nucleus?
-
 5:31
5:31
neutrons and atom stability
-
 0:24
0:24
latest image of an atom! 🔬
-
 16:23
16:23
3.6-hydrogenic atoms
-
 12:59
12:59
the modern model of the atom
-
 0:30
0:30
zooming into a water 💧
-
 0:18
0:18
what is the quantum model that describes electrons in atoms as occupying clouds of probability?
-
 0:32
0:32
how does an atom actually look like?
-
 2:02
2:02
voyage into the world of atoms
-
 13:42
13:42
chem143 electrons in atoms
-
 4:36
4:36
what do atoms look like?
-
 4:22
4:22
the simple atom - what causes an electron to orbit a proton?
-
 5:07
5:07
as-level chemistry: electrons in atoms part 1
-
 0:13
0:13
tamasha dekho 😂 iitian rocks relatives shock 😂😂😂 #jeeshorts #jee #shorts
-
 5:35
5:35
a better way to picture atoms
-
 26:28
26:28
calculating protons, neutrons, and electrons. orbital diagrams and electron configurations