getting to know changes of iso 14971 2019 risk management for medical devices
Published 4 years ago • 1.8K plays • Length 54:08Download video MP4
Download video MP3
Similar videos
-
 25:39
25:39
iso 14971 : 2019 ( medical device risk management ) | detailed explanation clause by clause
-
 35:48
35:48
medical device compliance with iec 62304 and iso 14971
-
 5:36
5:36
iso 14971:2019 – risk management for medical devices part 1
-
 16:03
16:03
what is new in iso 14971 2019
-
 52:32
52:32
iso 14971:2019 & tr 24971 explained - medical device risk management
-
 15:22
15:22
how to estimate risk for a medical device according to iso 14971:2019
-
 17:07
17:07
risk management for medical devices and iso 14971 - online introductory course
-
 11:10
11:10
5 key changes in iso14971:2019
-
 1:25:43
1:25:43
how to simplify your compliance with the new iso 13485:2016
-
 38:32
38:32
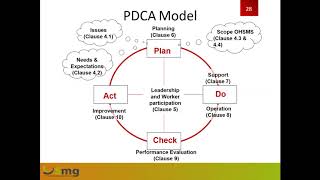
iso 45001:2018 occupational health and safety management system
-
 3:35
3:35
understanding iso 41001:2018: facility management system
-
 25:23
25:23
iso 14971: medical risk management best practices
-
 6:25
6:25
iso 14971 - understanding the term hazard
-
 3:28
3:28
how are iso 13485 and iso 14971 linked?
-
 2:45
2:45
mdr risk management training course - build, document & maintain an iso 14971:2019-compliant system
-
 1:35
1:35
mdr risk management templates - build an adaptive & iso 14971:2019-compliant risk management system
-
 1:20:58
1:20:58
what are the changes to iso 14971 2019? (replay) #medicaldevice
-
 1:34:48
1:34:48
webinar on “iso 14971:2019- tips to do better risk assessment on medical devices”
-
 8:19
8:19
live training: iso 14971:2019 risk management requirements
-
 10:28
10:28
fmea vs iso 14971
-
 1:12:06
1:12:06
medical devices - iso 14971 : risk management
-
 1:14
1:14
risk management webinar announcement (iso 14971/ce marking)