worked example: calculating solubility from kₛₚ | equilibrium | ap chemistry | khan academy
Published 3 years ago • 53K plays • Length 4:52Download video MP4
Download video MP3
Similar videos
-
 8:36
8:36
solubility product constant (ksp)
-
 41:52
41:52
ksp - molar solubility, ice tables, & common ion effect
-
 0:33
0:33
lanthanum iodate, la(io3)3, is a sparingly soluble salt with ksp = 7.5x10^-12. if 1200 ml of 0.0050…
-
 8:20
8:20
ksp and molar solubility calculations video
-
 2:33
2:33
saturated solution equilibrium (ksp) sample problem: chapter 17 – part 16
-
 13:50
13:50
complex ion equilibria - stepwise formation constant kf & ksp molar solubility problems
-
 5:34
5:34
molar solubility example calculation - common ion
-
 33:21
33:21
buffer solutions
-
 6:49
6:49
predicting precipitation with ksp values
-
 10:18
10:18
solubility product vs ionic product| precipitate formation|ksp vs ip|#chemistrycubicle
-
 3:18
3:18
⚗️ calculating molar solubility from kₛₚ (question 2)
-
 3:44
3:44
⚗️ predicting precipitation reactions by comparing q and ksp
-
 7:54
7:54
how to determine ksp given molar solubility
-
 1:10
1:10
solubility product (ksp) sample problem: chapter 17 – part 11
-
 2:42
2:42
another saturated solution equilibrium (ksp) sample problem: chapter 17 – part 18
-
 3:26
3:26
⚗️ calculating kₛₚ from molar solubility (ag₂so₄)
-
![[shortcut] how to solve for molar solubility given ksp](https://i.ytimg.com/vi/JKnSfMtykkQ/mqdefault.jpg) 4:51
4:51
[shortcut] how to solve for molar solubility given ksp
-
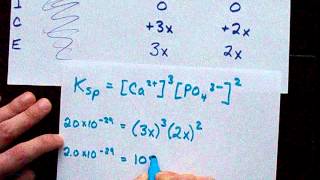 5:59
5:59
solubility equiliibrium (ice table and ksp)
-
 13:40
13:40
the `k_(sp)of ca(oh)_(2)is 4.42xx10^(-5)at 25^(@)c`. a 500 ml of saturated solution of `ca(oh)_(2)`
-
 2:00
2:00
ksp - solubility product constant molar solubility: example 1
-
 20:25
20:25
14.04 the solubility product constant