for equilibrium a(g) = b(g) c(g) at constant p of 1 atm, a dissociates 50 % at 500 k. find del go
Published 8 days ago • 12 plays • Length 5:34Download video MP4
Download video MP3
Similar videos
-
 3:12
3:12
sample problem 3.1 - using scientific notation
-
 0:33
0:33
algebra-based physics i (acts: phys2014) - corrected answer - problem set 7
-
 5:36
5:36
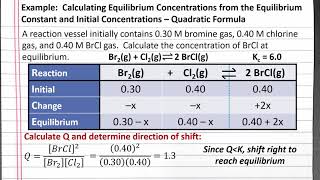
chem 201: calculating equilibrium concentrations from k and initial values – quadratic formula
-
 27:05
27:05
griffiths qm 2.2: infinite square well part 1: solving the well mathematically
-
 3:57
3:57
kc units || equilibrium constant units || kc calculations || a level chemistry
-
 1:06
1:06
nebvecoh
-
 11:20
11:20
kenneth brown's quantum primer
-
 1:16:49
1:16:49
quantum machine learning beyond kernel methods
-
![[ib math aa hl] [2023/may/tz1/p3/q2]](https://i.ytimg.com/vi/ZPYU90bjVj0/mqdefault.jpg) 14:01
14:01
[ib math aa hl] [2023/may/tz1/p3/q2]
-
 1:01:35
1:01:35
robert huang: fundamental aspects of solving quantum problems with machine learning
-
 19:37
19:37
homin jiang - development of 16 gb per sec board
-
 17:24
17:24
(iwqc20) quanlong wang: exact synthesis of quantum circuits in algebraic zx-calculus
-
 4:12
4:12
#1 - calculations with scientific notation | addition and subtraction
-
 4:26
4:26
quantum chemistry 1.0 - early quantum review
-
 2:30
2:30
quantum scientists in singapore: kuan eng johnson goh
-
 2:54
2:54
quantum scientists in singapore: ng hui khoon
-
 4:24
4:24
kb
-
 1:27
1:27
1m25b
-
 11:02
11:02
ka, kb, kw
-
 45:57
45:57
2021.07.13, jaehoon kim (김재훈), k_{r 1}-saturated graphs with small spectral radius
-
 2:11
2:11
korea's central bank keeps key rate steady at 1.5% in may