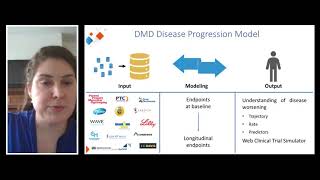
2022 rdca-dap workshop: case study 3, duchenne regulatory science consortia (d-rsc)
Published 2 years ago • 227 plays • Length 24:22Download video MP4
Download video MP3
Similar videos
-
 1:16:37
1:16:37
2022 rdca-dap workshop: future of rdca-dap and the patient voice
-
 41:33
41:33
2022 rdca-dap workshop: new collaborations for rdca-dap
-
 1:36:06
1:36:06
webinar: rdca-dap and eatris present rare disease drug development
-
 11:26
11:26
2022 rdca-dap workshop: case study 2, critical path for sickle cell disease consortium (cp-scd)
-
 58:49
58:49
webinar: how rdca-dap can help inform optimal trial design in progressive rare disease
-
 48:02
48:02
rdca-dap - value of integrated data and analytics in rare disease drug development
-
 26:47
26:47
2022 rdca-dap workshop: centrality of patient advocacy organizations in accelerating treatments
-
 2:38
2:38
global burden of disease neck pain @kiropraktorerik
-
 1:04:57
1:04:57
cgic2024 day 1: ai algorithm literacy
-
 29:19
29:19
end to end regulatory information management case study of a veeva rim implementation
-
 25:53
25:53
2021 rdca-dap annual workshop: examples of multiple datasets shared within one disease group
-
 7:01
7:01
2022 rdca-dap workshop: opening remarks
-
 15:20
15:20
2022 rdca-dap workshop: keynote
-
 12:03
12:03
rdca-dap drug development tool prototypes
-
 1:19:08
1:19:08
2022 rdca-dap workshop: impact of rdca-dap panel discussion
-
 55:57
55:57
sharing data with rdca-dap
-
 38:32
38:32
rdca-dap launch live stream - am part 1
-
 1:25:50
1:25:50
rdca-dap launch live stream - pm part 1
-
 4:16
4:16
rdca-dap platform preview
-
 8:03
8:03
rdca-dap: how the data and analytics platform accelerates rare disease drug development
-
 1:51
1:51
rare disease patient voice: justin moy