managing regulatory submission documentation for life sciences
Published 4 years ago • 879 plays • Length 2:39Download video MP4
Download video MP3
Similar videos
-
 1:31
1:31
simplify planning and assembly of regulatory submissions for life sciences
-
 2:47
2:47
managing essential clinical trial documentation for life sciences
-
 2:17
2:17
managing quality and manufacturing documentation for life sciences
-
 47:21
47:21
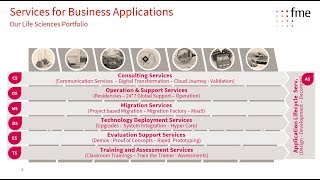
opentext documentum life sciences solution suite #4 submission store & view
-
 2:19
2:19
working with a document | open text documentum for life sciences
-
 1:09:32
1:09:32
regulatory documents explained - dhf, dmr, dhr and tf
-
 47:28
47:28
risk based validation of laboratory systems recording 02092012
-
 4:11
4:11
📖📑📕💿ctd, nees & ectd preparation & submission ( asmaa khalil )
-
 2:09
2:09
opentext life sciences solutions
-
 2:00
2:00
industry-leading life sciences solutions in the cloud
-
 35:32
35:32
a complete submission publishing solution for life sciences 2022 demo
-
 8:53
8:53
integrating your ecm and regulatory submissions - part 1
-
 2:15
2:15
create a submission | core for regulatory plans
-
 12:31
12:31
a flexible way to support regulatory, quality and clinical users using life sciences smart view
-
 54:59
54:59
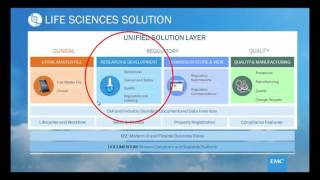
opentext documentum life sciences solution suite #3 research and development 2017-10-19
-
 39:21
39:21
45 minutes on emc documentum research & development & submission store view
-
 55:56
55:56
opentext documentum life sciences solution suite #5 electronic trial master file 2018-01-18
-
 1:17
1:17
how to delete and restore a document | opentext documentum for life sciences
-
 49:33
49:33
ctn webinar: irb and regulatory documentation.
-
 2:06
2:06
opentext™ life sciences smart view overview
-
 30:47
30:47
emc documentum for life sciences solution suite 4.0 – 30 min webcast on electronic trial master file
-
 1:36
1:36
regulation management for life sciences