mdic framework launch: closing remarks from fda deputy commissioner rob califf
Published 9 years ago • 56 plays • Length 20:25Download video MP4
Download video MP3
Similar videos
-
 19:16
19:16
mdic framework launch: catalog overview
-
 18:14
18:14
mdic framework launch: framework overview - ross jaffe
-
 52:52
52:52
mdic framework launch: cdrh perspectives on patient preferences
-
 30:12
30:12
2016 mdic annual public forum; keynote, dr. robert califf
-
 33:47
33:47
mdic live with case for quality
-
 12:18
12:18
where to find fda software and cybersecurity submission requirements
-
 8:34
8:34
what is breakthrough device designation?
-
 1:34:30
1:34:30
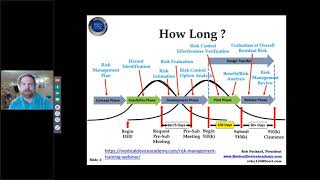
how to prepare a medical device 510k submission for fda | rob packard | joe hage | updated
-
 1:27
1:27
investigational device exemption
-
 28:50
28:50
closing keynote: cdrh updates
-
 45:56
45:56
nest forum closing session
-
 1:42
1:42
breakthrough device designation | proxima cro
-
 1:04
1:04
international medical device regulators forum (imdrf) australia 2022 launch.
-
 1:01
1:01
how did we #makecapacool?
-
 2:16
2:16
breaking down the fda pre-submission process - an essential guide
-
 2:45
2:45
changes to the fda ecopy submission process
-
 2:01:43
2:01:43
case for quality forum - morning session
-
 2:06
2:06
fda reviews medical device approval process