medical device registration in the us- the 510(k)-application process
Published 3 years ago • 584 plays • Length 3:21Download video MP4
Download video MP3
Similar videos
-
 2:24
2:24
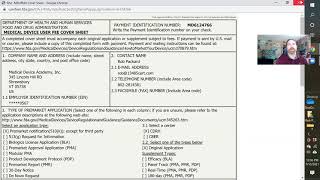
medical device registration in the us - the pma application process
-
 2:29
2:29
medical device registration in the us- the de novo request application process
-
 4:36
4:36
medical device registration in the us- post-approval compliance requirements.
-
 4:46
4:46
medical device registration in the us- an overview of pathways and classification
-
 21:10
21:10
how to register a medical device with fda? (510k, pma, de novo...)
-
 1:36:15
1:36:15
software validation documentation for fda 510(k) pre-market notification submission
-
 46:31
46:31
fda cybersecurity testing requirements - interview with red sentry
-
 12:18
12:18
where to find fda software and cybersecurity submission requirements
-
 4:07
4:07
how to register a medical device in the usa (ide)
-
 2:33
2:33
medical device regulations in malaysia
-
 3:59
3:59
united states medical device registration chapter 1 - overview
-
 33:03
33:03
how to register a medical device through fda 510k with michelle lott
-
 5:13
5:13
united states medical device registration chapter 5 - dossier preparation
-
 3:13
3:13
medical device registration in brazil - part i - the role of anvisa and brh
-
 0:56
0:56
freyr has successfully obtained approvals for 100 medical devices & ivds
-
 3:25
3:25
united states medical device registration chapter 3 - quality management system
-
 14:46
14:46
u s fda medical device pre market regulatory submissions
-
 5:07
5:07
u.s. fda’s 510(k), ide, and pma documentation, submission and approval process
-
 9:28
9:28
medical device regulations / fda approval
-
 11:06
11:06
how to prepare a medical device 510k submission for fda
-
 22:34
22:34
fda establishment registration and listing for medical devices
-
 15:04
15:04
cybersecurity documentation for a 510(k) submission