operational qualification: qualified vs. validated limits | medtech
Published 1 year ago • 137 plays • Length 0:27Download video MP4
Download video MP3
Similar videos
-
 1:07
1:07
what is an operational qualification (oq) in medtech?
-
 1:13
1:13
operational qualification – how many samples do you need? | medtech
-
 1:28
1:28
operational qualification: equipment oq vs. process oq | medtech
-
 0:40
0:40
where to get the limits for the operational qualification (oq) runs from?
-
 0:37
0:37
does an operational qualification (oq) always have 2 runs? | medtech
-
 0:45
0:45
checklist operational qualification (oq) – what you need to start!
-
 1:37:49
1:37:49
9. verification and validation
-
 1:28:00
1:28:00
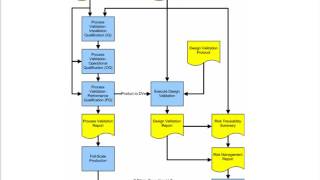
process validation for medical device manufacturers
-
 57:08
57:08
an introduction to iec 62304 - software for active medtech
-
 0:39
0:39
when to validate processes in medtech?
-
 0:34
0:34
process validation requirements: is periodic re-validation required in medtech?
-
 0:41
0:41
do i have to validate manual processes in medical technology?
-
 1:26
1:26
when do i need to perform installation qualification when manufacturing medical devices?
-
 0:48
0:48
what is an installation qualification (iq)? | medical device production
-
 0:55
0:55
test method validation standards and guidelines | medical devices
-
 0:59
0:59
where can i find the requirements for process validation?
-
 0:44
0:44
installation qualification – where to find the requirements? | medtech
-
 1:05
1:05
performance qualification (pq) in medtech – how many runs do you have to produce for a pq?
-
 0:30
0:30
installation qualification checklist – where to get information from?