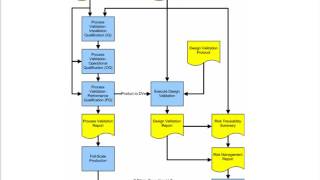
protocols for medical devices & process validation principles
Published 8 years ago • 5.8K plays • Length 1:08:12Download video MP4
Download video MP3
Similar videos
-
 1:08:12
1:08:12
process validation principles and protocols for medical devices
-
 1:28:00
1:28:00
process validation for medical device manufacturers
-
 1:28:00
1:28:00
process validation procedure for medical device manufacturers
-
 6:35
6:35
sys-014 process validation procedure
-
 12:49
12:49
process validation for medical devices - short course
-
 8:56
8:56
process validation or verification for your medical device (iso 13485)
-
 1:01:22
1:01:22
packaging validation of medical devices - impact of the revisions of iso 11607 & suitable strategies
-
 1:36:15
1:36:15
software validation documentation for fda 510(k) pre-market notification submission
-
 1:18:50
1:18:50
statistical concepts of process validation
-
 4:46
4:46
process validation protocols & reports 820.75 & iso 13485 § 7.5.6 (executive series #66)
-
 38:14
38:14
how to perform your process validation for medical devices? (iq oq pq)
-
 1:16:37
1:16:37
cgmp for medical devices including ivds
-
 8:50
8:50
process validation | types of process validation | process performance qualification
-
 59:53
59:53
medical device design control