re-classification of software based medical devices
Published 3 years ago • 478 plays • Length 1:23Download video MP4
Download video MP3
Similar videos
-
 59:50
59:50
tga webinar: regulation of software as a medical device
-
 24:59
24:59
webinar: sme assist - personalised medical devices framework
-
 1:06:30
1:06:30
2021-09-08 working with the tga
-
 1:43
1:43
tga regulatory compliance
-
 52:53
52:53
application for consent to import, supply, or export a medical device non-compliant with the ep
-
 15:37
15:37
what’s in attachment g of the new fda biocompatibility guidance?
-
 29:25
29:25
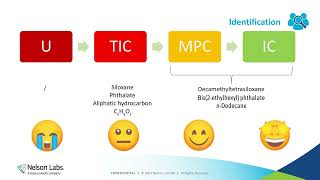
medical device chemical characterization
-
 9:17
9:17
mobile forensics tools - hardware
-
 8:41
8:41
a summary of supplying therapeutic goods in australia
-
 1:02:31
1:02:31
tga medical device changes in australia
-
 53:03
53:03
unique device identification webinar #13 - udi consultation paper 3 information and project update
-
 54:53
54:53
tga update on medical devices february 2019
-
 15:35
15:35
what is a de novo?
-
 2:37
2:37
australian tga medical device regulations | qreg | medical device consulting services
-
 51:35
51:35
application to import, supply or export non-compliant medical devices
-
 0:45
0:45
snapshot - therapeutic goods administration
-
 1:45
1:45
discover rams - medical device regulatory intelligence software
-
 5:43
5:43
what is software as a medical device (samd) in clinical research?
-
 5:48
5:48
what is a medical device (per the eu medical device regulations)?
-
 32:08
32:08
medical device regulatory podcast - the journal of medical device regulation
-
 7:07
7:07
regulatory affairs update - australia - nov 2021
-
 38:45
38:45
medical device software: current developments in the regulatory world