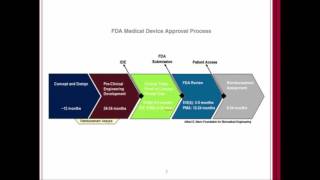
regulatory pathways of medical devices in usa and european union
Published 3 years ago • 1.2K plays • Length 7:13Download video MP4
Download video MP3
Similar videos
-
 4:35
4:35
what everybody should know about clinical trials! - part 2 - medical device legislation
-
 5:58
5:58
good clinical practice and ich gcp guidelines
-
 4:49
4:49
regulatory challenges on medical device studies - challenges in clinical research
-
 10:36
10:36
regulatory authority inspections in clinical trials - part 1
-
 13:14
13:14
clinical evaluation of medical devices prior and after mdr
-
 4:15
4:15
why should drug-device combination manufacturers know the different regulations in the usa and eu?
-
 5:59
5:59
medical device classes
-
 13:18
13:18
everything you need to know about the ivdr
-
 28:43
28:43
gcp mindset: modern quality assurance
-
 15:54
15:54
fda regulation of medical devices and software/apps
-
 16:55
16:55
how to update your clinical evaluation report (cer) for 2024
-
 18:46
18:46
medical devices: summary of safety and clinical performance
-
 5:28
5:28
what is regulatory compliance in clinical trials?
-
 5:21
5:21
patient reimbursement for clinical trials in the eu
-
 9:33
9:33
the role of the person responsible for regulatory compliance (prrc) in medical device regulation
-
 6:38
6:38
inspection readiness of the tmf
-
 3:32
3:32
what everybody should know about clinical trials! - part 3 - what is a medical device?
-
 4:44
4:44
why the development of in vitro diagnostics is different?
-
 7:46
7:46
understanding mdcg 2023-7: exemptions from clinical investigations for medical devices
-
 7:31
7:31
cra basics: what is risk-based monitoring in clinical research?
-
 7:18
7:18
navigating the cip process in medical device clinical trials